When someone hands you a vial of cells and says “there are 100 million cells in there,” it sounds like a complete answer. It sounds precise, even scientific. But that number, on its own, tells you surprisingly little about what you actually have in that vial. The real story depends on how many of those cells are alive, what type of cells they are, whether the live ones are actually capable of doing anything useful, and a dozen other variables that a simple count will never capture.
This matters more than most people realize. In clinical settings, a miscommunication about viable cell dose can mean the difference between a treatment working and it failing entirely. In research, it can make experiments impossible to reproduce. In the growing world of consumer-facing cell therapies, including clinics offering stem cells in Colombia and elsewhere across Latin America, it can mean that patients pay significant sums of money for products that have far less biological value than advertised. Understanding what a cell count actually tells you, and what it does not, is not just an academic exercise. It is genuinely important.
What a Cell Count Measures and What It Leaves Out
A cell count is exactly what it sounds like: a number that represents how many cells are present in a given volume. But even that simple definition hides complexity. Are you counting all cells, including dead ones? Only nucleated cells? Only a specific cell type within a mixed population? The answer changes the number dramatically, and different labs and manufacturers use different definitions without always making that clear.
What a cell count does not tell you is whether those cells are alive. It does not tell you whether the live cells are healthy and functional. It says nothing about whether you have the right type of cells for your intended purpose. And it gives no information about whether the cells were stressed, damaged, or compromised at any point during collection, processing, or storage. In other words, a cell count is the starting point for understanding what you have, not the finish line.
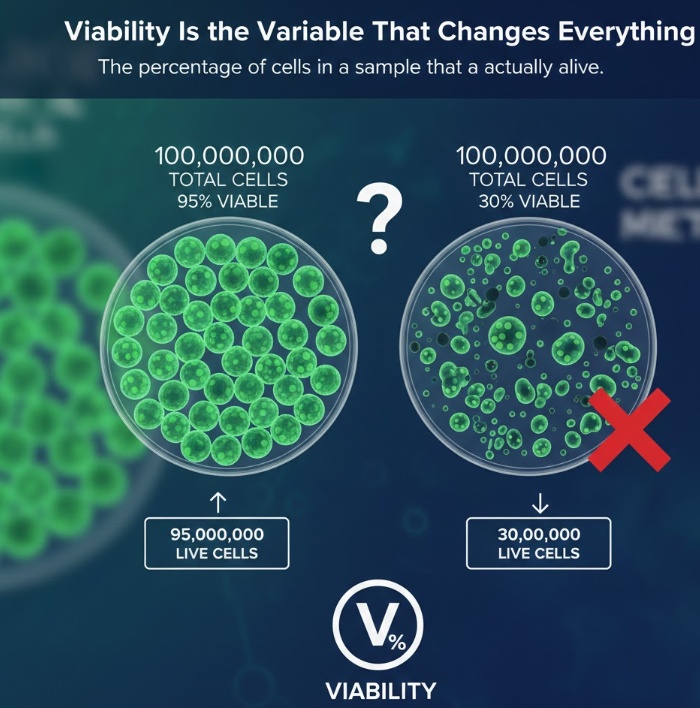
Viability Is the Variable That Changes Everything
Viability refers to the percentage of cells in a sample that are actually alive. It is reported as a percentage, and it completely transforms what a raw cell count means in practice.
Consider two vials, both labeled “100 million cells.” One has a viability of 95 percent. The other has a viability of 55 percent. The first vial contains roughly 95 million live cells. The second contains about 55 million. That is a gap of 40 million living cells between two products that appear identical on a label. If you are a researcher trying to set up a reproducible experiment, or a clinician trying to deliver a therapeutic dose, that difference is enormous.
Viability degrades for a number of reasons. The longer cells sit after collection, the more of them die off. Temperature fluctuations during transport can be lethal, especially for sensitive cell types. Freeze-thaw cycles cause physical damage to cell membranes. Processing steps like centrifugation and density gradient separation, while necessary, apply mechanical and osmotic stress that pushes some cells toward death. In short, by the time cells go from a donor to a lab bench or a patient, the original cell count may bear little resemblance to the number of viable cells remaining.
Despite all of this, it is common for manufacturers and suppliers to report only the raw cell count. Viability is often not listed at all, or is buried in a secondary document that most customers never see. In some cases, products with poor viability continue to circulate because sellers either do not test for it rigorously or do not consider it a primary selling point.
Why the Counting Method Matters More Than You Think
You might assume that counting cells is a standardized process, that a count from one lab should match a count from another lab for the same sample. That is not always true. The method used to count cells affects the result, sometimes significantly.
The most traditional method is the hemocytometer combined with Trypan Blue exclusion. This dye enters cells that have lost membrane integrity, staining them blue and marking them as dead. Live cells with intact membranes exclude the dye and remain clear. A technician then counts the colored and uncolored cells under a microscope. This works reasonably well, but it introduces human error, takes time, and suffers from an important limitation: it cannot distinguish between cells that are truly alive and healthy and cells that are in the early stages of dying. Early apoptotic cells, meaning cells that have already begun the programmed cell death process, often still have intact membranes at the time of counting. They will appear as “live” under Trypan Blue but are functionally useless or worse.
Automated cell counters improve reproducibility and reduce human error, but they rely on the same basic dye exclusion principle and carry the same blind spot for early apoptosis. Flow cytometry-based counting is more sophisticated. It can simultaneously assess membrane integrity, cell type identity, and apoptotic status using multiple markers at once. It provides a far more accurate picture of what is actually alive and functional in a sample, but it is also more expensive and requires specialized equipment and expertise.
The practical implication is that the same physical sample can generate genuinely different cell counts depending on the method used. A Trypan Blue count might read 90 million live cells in a sample that, when assessed by flow cytometry with apoptosis markers, turns out to contain only 70 million cells that are truly healthy and functional. Both numbers are “accurate” given their respective methods. Neither number is wrong. But they are telling you different things, and if you do not know which method was used, you cannot properly interpret the number you are given.
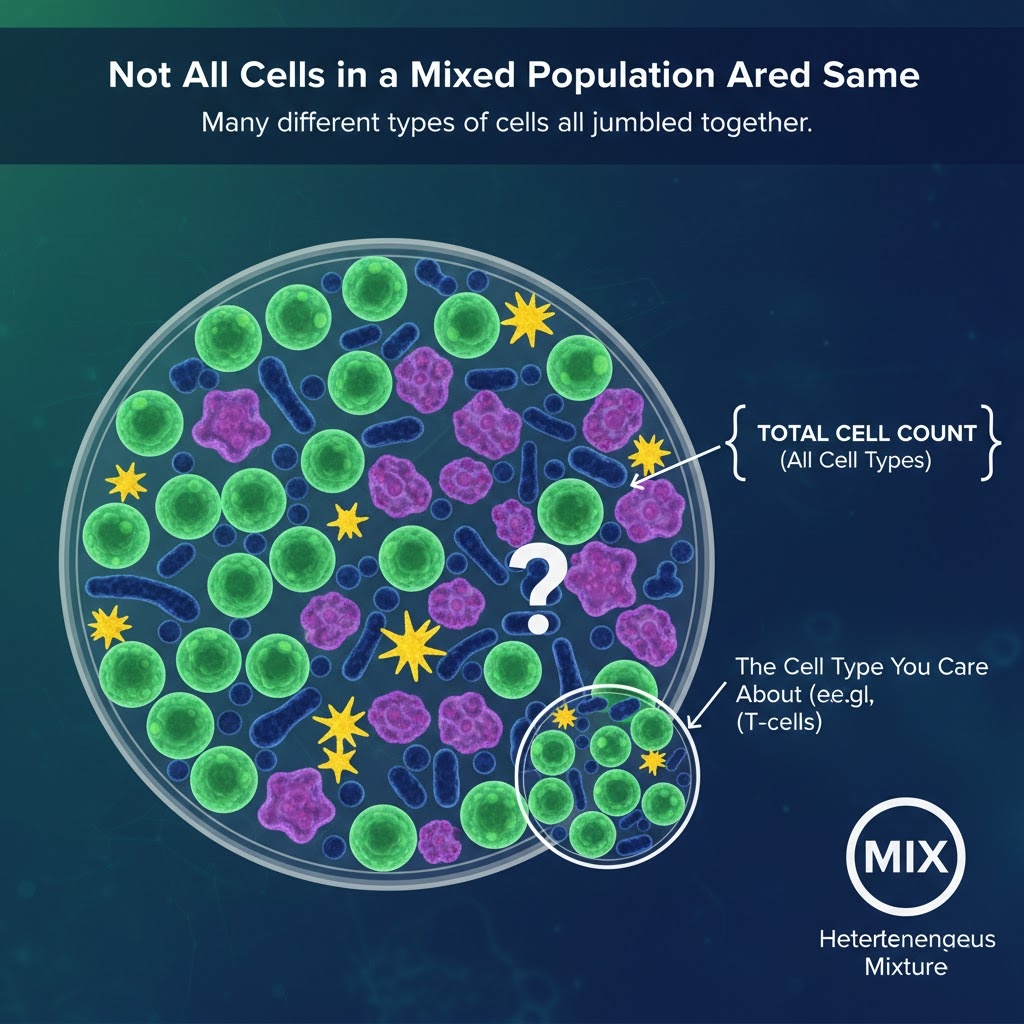
Not All Cells in a Mixed Population Are the Same
Many cell products are not pure populations of a single cell type. They are heterogeneous mixtures, meaning they contain many different types of cells all jumbled together. Peripheral blood mononuclear cells, cord blood units, and bone marrow aspirates are common examples. When you count cells in these kinds of samples, you get a total number that represents all cell types present, not just the one you care about.
A cord blood unit might contain 100 million total nucleated cells. But the cells that actually matter for a bone marrow transplant are the CD34-positive hematopoietic stem and progenitor cells, which are present in much smaller quantities. Two cord blood units with identical total cell counts might contain very different numbers of CD34-positive cells depending on donor biology and collection technique. The unit that looks equivalent by total count might be clinically inferior, or even unsuitable for a given patient, when you look at the relevant subset.
This is not an abstract concern. Transplant physicians do not use total cell counts to select cord blood units for their patients. They look at CD34-positive cell counts and other markers of engraftment potential. The total count is essentially irrelevant to their decision. Yet total cell counts are the number most commonly used in public-facing communications, in banking agreements, and in marketing materials.
Phenotypic drift is another factor that rarely gets discussed. Cells change their identity over time and with repeated passaging in culture. A mesenchymal stem cell at passage 3 and the same cell at passage 10 can have meaningfully different gene expression profiles and functional properties, even if they count up the same under a microscope. The label on the vial may not have changed, but the contents have.
Being Alive Is Not the Same as Being Effective
This is the part of the discussion that is most routinely skipped, even by people who are careful about viability. A cell can be alive by every standard measure and still be completely ineffective at doing what you need it to do.
Cellular senescence is a good example. Senescent cells have stopped dividing and have entered a state of permanent growth arrest. They are metabolically active and will look fine under a Trypan Blue assay. They will count as “live.” But they are no longer capable of proliferating, regenerating tissue, or carrying out the specialized functions that make them therapeutically useful. Depending on the cell type and the application, a batch of cells that is 80 percent viable but heavily senescent may deliver far less biological effect than a batch that is 70 percent viable but composed of young, actively functional cells.
Functional exhaustion is another issue, common in immune cell therapies. T cells and natural killer cells that have been over-stimulated or improperly maintained during manufacturing can enter an exhausted state where they are alive but unresponsive. They cannot mount an effective immune attack even when exposed to their target. This is a real clinical problem in some CAR-T cell therapies and is an area of ongoing research.
This is why potency assays exist. Rather than simply counting cells or checking membrane integrity, a potency assay asks whether the cells can actually do their job. For stem cells, colony-forming unit assays test whether a cell can give rise to new colonies of daughter cells. For natural killer cells, cytotoxicity assays measure their ability to kill target cells. For mesenchymal stem cells, cytokine secretion profiles can indicate whether the cells are secreting the anti-inflammatory and regenerative signals they are expected to provide. These assays add cost and complexity, but they are the only way to know whether a viable cell count translates into a potent, clinically useful product.

The Gap Between Pre-Freeze and Post-Thaw Numbers
A large proportion of cell products are cryopreserved, meaning they are frozen and stored until needed. Cryopreservation is practical and necessary, but it is not free. Cells take damage during freezing and thawing, and that damage reduces viability.
When cells are frozen, ice crystals can form both inside and outside the cell. Even with cryoprotectants like DMSO, which help prevent ice crystal formation, the process causes osmotic stress, membrane disruption, and sometimes direct physical damage. When cells are thawed, additional damage can occur if the process is not handled correctly. The net result is that a cell product that went into the freezer at 95 percent viability may come out with a viability of 70 to 80 percent, or lower, depending on how it was processed, what freezing protocol was used, and how long it was stored.
The standard practice in much of the industry is to report the pre-freeze cell count and viability. This is the number measured before the product goes into the freezer. It is also the highest number the product will ever have. The post-thaw count, which is the number that actually matters for the end user, is often not reported or is reported only on request.
This is a significant transparency problem. A cord blood bank might advertise a unit as containing 100 million cells at 92 percent viability. Those are the pre-freeze numbers. After thawing, that unit might contain 75 million cells at 75 percent viability. The difference is substantial, and a transplant physician working from the pre-freeze numbers may be making decisions based on a product that no longer exists in that form. The push for mandatory post-thaw viability reporting has been gaining ground in regulatory discussions, but it has not yet become universal practice.
Processing Steps That Quietly Reduce Your Cell Dose
Before a cell product ever reaches a freezer or an end user, it goes through a series of processing steps. Each of those steps introduces the potential for cell loss. Most people who receive a cell product have no idea how many steps it went through or how each step affected the final yield.
The time between collection and processing is one of the most significant variables. Cells begin dying as soon as they are removed from their natural environment. A cord blood unit or bone marrow aspirate that sits for six hours before processing will arrive with meaningfully lower viability than one processed within two hours of collection. Temperature during transport and storage compounds this. Cells maintained at 4 degrees Celsius will generally survive longer than cells kept at room temperature, but even cold storage is not neutral.
The processing itself introduces additional losses. Centrifugation applies physical force that damages fragile cells. Density gradient separation, which is used to isolate specific cell populations from a mixed sample, involves layering cells over a density medium and spinning them down. It is effective but not gentle. Each washing step, each buffer exchange, and each volume reduction procedure removes some additional fraction of the cell population. In a well-run GMP facility with tight protocols, these losses are minimized and predictable. In less controlled environments, they can be substantial and variable. When you receive a vial of cells at the end of this chain, there is typically no way to know how much attrition occurred along the way.
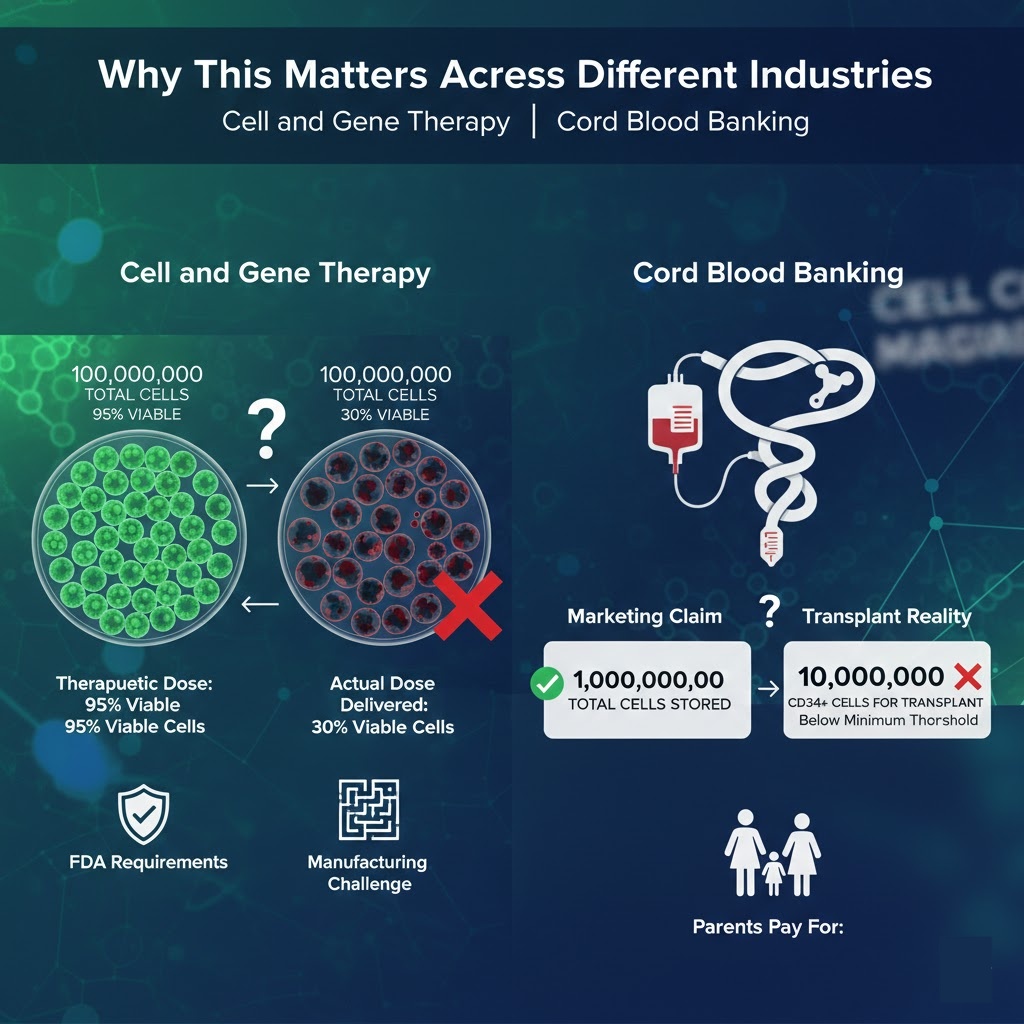
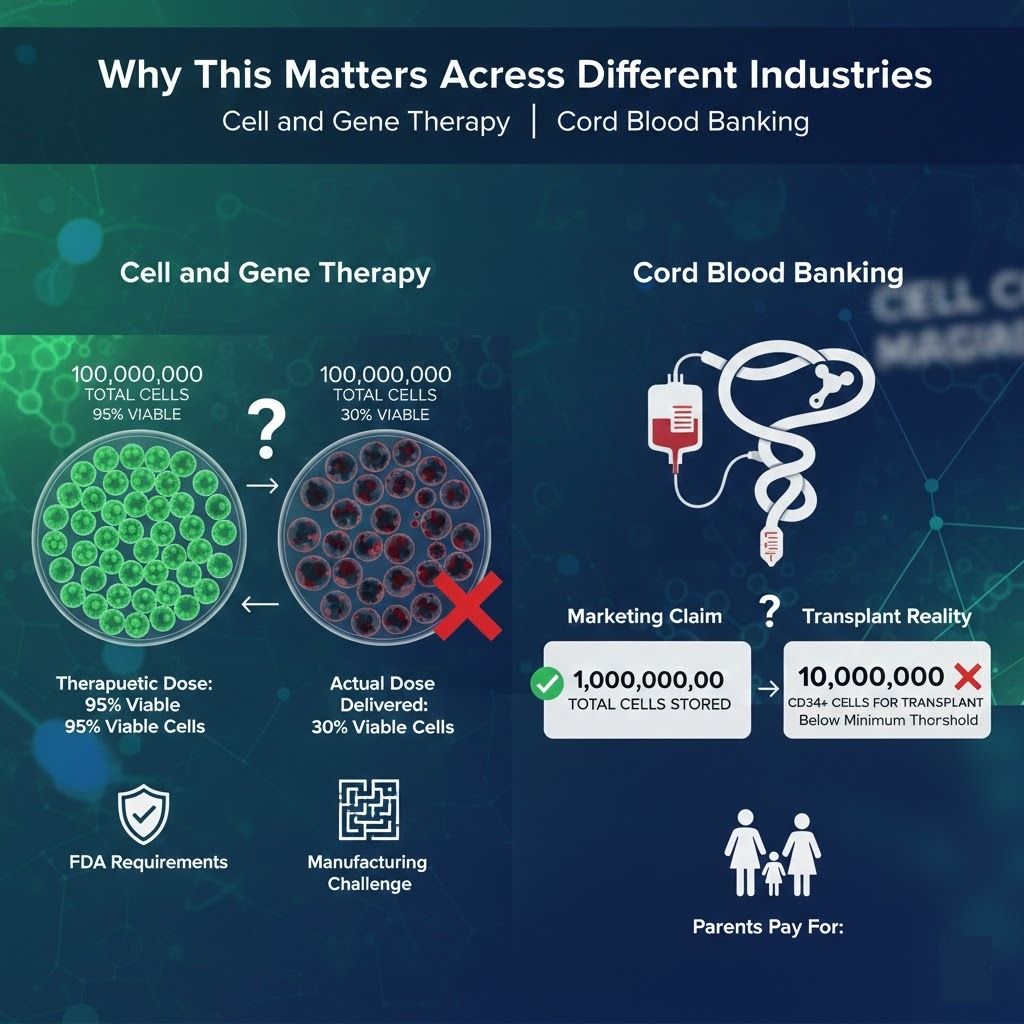
Why This Matters Across Different Industries
Cell and Gene Therapy
In cell and gene therapy, dosing precision is directly tied to patient outcomes. If a patient receives significantly fewer viable, potent cells than the therapeutic dose calls for, the treatment may simply not work. The FDA and other regulatory agencies require detailed potency and viability data as part of the approval process for these products, and release criteria typically include both viability thresholds and functional assessments. Even so, the gap between what is required on paper and what is consistently delivered in practice remains a challenge, particularly for smaller manufacturers.
Cord Blood Banking
Cord blood banking may be where the cell count communication problem is most visible to ordinary consumers. Parents banking their newborn’s cord blood are paying for a service that is marketed heavily around the total cell count collected and stored. Marketing materials often emphasize large numbers without explaining viability, post-thaw performance, or the CD34-positive subset counts that actually determine whether a unit is useful for transplant. Many stored units fall below the minimum thresholds that transplant programs require, a fact that is rarely communicated clearly at the time of banking.
Research and Academic Labs
In academic research, inconsistent cell counting practices contribute to reproducibility problems that have frustrated the scientific community for years. If one lab counts cells using a hemocytometer with a loose counting protocol and another uses automated counting with strict viability thresholds, the starting conditions for experiments labeled as identical can be quite different. Mycoplasma contamination is a particularly insidious issue because it degrades cell viability and function without producing obvious visual signs of contamination. Cells in an infected culture can look normal under a microscope while performing poorly in functional assays.
Direct-to-Consumer Stem Cell Clinics
The direct-to-consumer stem cell clinic space presents unique challenges around cell count transparency. Some clinics advertise treatments using cell counts as the primary measure of product quality and therapeutic value. A large cell count sounds impressive and reassuring to a patient who does not know what questions to ask. But without accompanying viability data, post-thaw performance, cell type characterization, and potency information, a large number is essentially a marketing figure rather than a clinical one.
What Regulators Require and Where the Gaps Are
Regulatory agencies have become increasingly sophisticated in how they think about cell products. The FDA’s regulations under 21 CFR Part 1271 govern human cells, tissues, and cellular and tissue-based products. For products subject to a biologics license application or an investigational new drug filing, the agency requires detailed characterization including viability, identity, purity, and potency. The International Society for Cell and Gene Therapy has published guidelines emphasizing potency as a required quality attribute, not an optional one.
Internationally, accreditation bodies like FACT and NETCORD set standards for cord blood banks that include minimum viable cell thresholds and specific requirements around CD34-positive cell counts. The ISBT 128 labeling standard is designed to ensure that units carry accurate, standardized information across the transplant system.
The gaps, however, are real. For cell products that are not subject to full biologics licensing, particularly those that fall under the agency’s enforcement discretion policies for certain same-day surgical procedures, characterization requirements are much lighter. And for products sold in markets with less rigorous oversight, a manufacturer may face no binding requirement to report post-thaw viability, cell type composition, or any measure of potency at all. The number on the label may be technically accurate as a total cell count while communicating almost nothing about the actual quality of the product.
What Good Reporting Actually Looks Like
There is a clear set of practices that separate high-quality cell product reporting from bare-minimum reporting. Viability should always be reported alongside a cell count, not as a footnote but as an equally prominent figure. The counting method should be specified so that the recipient can properly interpret the number. Where the product is a heterogeneous mixture, the relevant cell subtype counts should be disclosed, not just the total.
For cryopreserved products, post-thaw data should be provided as a matter of course. This is the number that actually describes what the end user will receive, and it should be the primary figure in any product documentation. A potency or functional assessment, appropriate to the cell type and intended use, should accompany the viability data wherever possible.
Timing matters too. Cell viability degrades after thawing, and a count taken immediately after thaw is different from one taken two hours later. Time-stamping counts and specifying the conditions under which they were taken is a best practice that is straightforward to implement but often skipped.
Finally, the full processing history of a product should be documented and accessible. How long did the material sit before processing? What separation technique was used? How many freeze-thaw cycles has it gone through? This information allows the end user to make an informed judgment about the product they are receiving.
Questions Anyone Receiving Cell Products Should Ask
Whether you are a clinician selecting a cord blood unit, a researcher ordering cell lines, or a patient considering a cell therapy, there are specific questions that can help you understand what you are actually getting.
Ask for the post-thaw viability, not just the pre-freeze numbers. Ask what counting method was used and whether an apoptosis marker was included. Ask for the CD34-positive count or other relevant subset counts if you are dealing with a mixed cell population. Ask whether a potency assay was performed and, if so, what the result was. Ask when the count was taken relative to the most recent thaw.
When reviewing a certificate of analysis, look for whether viability is reported as a percentage or a raw number, and check whether the assay method is specified. A CoA that lists only total cell count with no viability data is a significant warning sign. One that reports pre-freeze numbers without post-thaw follow-up is another. Marketing materials that emphasize cell counts without any other quality metrics should be read with appropriate caution.
The difference between a genuinely useful cell product and a disappointing one often lies not in the cells themselves but in how well the manufacturer has characterized and communicated what they actually have. Asking the right questions is the most direct way to tell the difference.
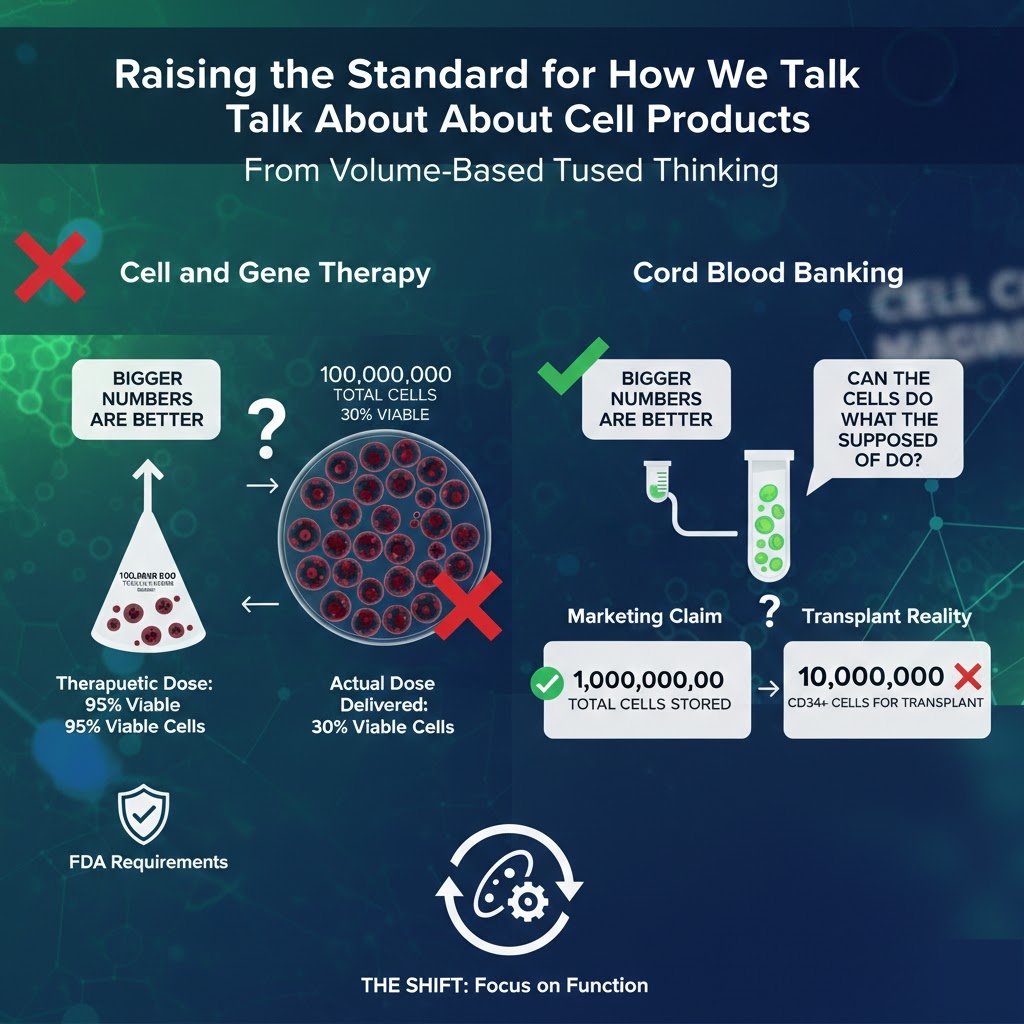
Raising the Standard for How We Talk About Cell Products
The shift that needs to happen in this field is conceptually simple even if it is practically challenging. The industry needs to move away from volume-based thinking, where bigger numbers are inherently better, and toward function-based thinking, where what matters is whether the cells can actually do what they are supposed to do.
A vial containing 60 million viable, potent, well-characterized cells is more valuable than one containing 150 million cells of uncertain viability, mixed phenotype, and unknown functional status. That should be obvious, but the current norms around how cell products are reported and marketed make it easy to obscure. Standardizing around post-thaw viability, subset characterization, and functional potency data would go a long way toward giving everyone in the chain, from manufacturers to clinicians to patients, the information they need to make good decisions.
Cell counts are useful. They are not meaningless. But they are the beginning of the characterization conversation, not the end of it. A number without context is just a number, and in cell biology, context is everything.