Every year, millions of umbilical cords are delivered alongside newborn babies and then thrown away as medical waste. For most of human history, that made sense. The cord had done its job, the baby was healthy, and there was nothing more to think about. But over the last few decades, scientists have discovered that one particular component of the umbilical cord contains something that may turn out to be one of the most medically valuable biological materials ever identified. That material is Wharton’s Jelly, and the stem cells it contains are changing how researchers and clinicians think about regenerative medicine.
The claim that Wharton’s Jelly-derived mesenchymal stem cells (MSCs) are the “gold standard” among all MSC sources is not marketing language. It reflects a growing body of peer-reviewed research showing that these cells outperform their competitors in several important categories. Understanding why requires a closer look at what Wharton’s Jelly actually is, what MSCs do, and what the science says about why the combination of the two is so compelling.
What Is Wharton’s Jelly
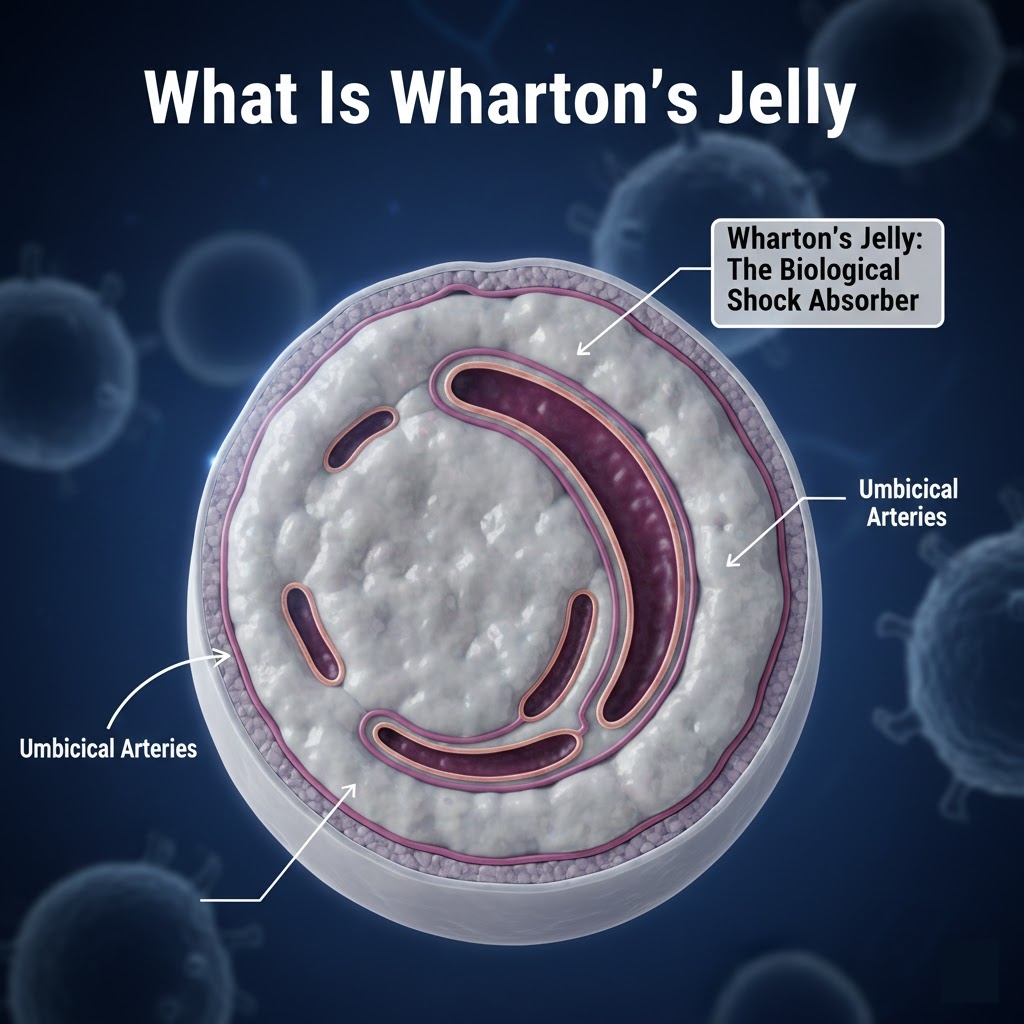
Wharton’s Jelly is the gelatinous connective tissue that fills the interior of the umbilical cord. Its primary job during fetal development is to protect the umbilical arteries and vein from compression, twisting, and mechanical stress. Think of it as a biological shock absorber. Without it, the blood vessels inside the cord would be far more vulnerable to the movements of the fetus and the pressures of the uterine environment.
The tissue was first described in 1656 by English anatomist Thomas Wharton in his work “Adenographia.” At the time, Wharton simply noted its existence as a structural component of the cord. It would be several centuries before anyone suspected it held something more interesting than mucus and collagen.
At a compositional level, Wharton’s Jelly is made up of a hydrated extracellular matrix (ECM) that is rich in hyaluronic acid, various types of collagen (primarily types I, III, and IV), and proteoglycans. Embedded within this matrix are stromal cells that, when properly isolated and cultured, display all the hallmark characteristics of mesenchymal stem cells. What makes these cells particularly interesting from a biological standpoint is that they come from a very primitive tissue. Wharton’s Jelly forms early in fetal development and remains relatively sheltered from the kinds of environmental stressors that age cells over time. The cells inside it have never been exposed to decades of inflammation, oxidative stress, or immune system activation. In a very real sense, they are among the youngest cells a researcher could ever work with.
What Mesenchymal Stem Cells Actually Are
Before getting into what makes Wharton’s Jelly MSCs special, it helps to understand what MSCs are in the first place.
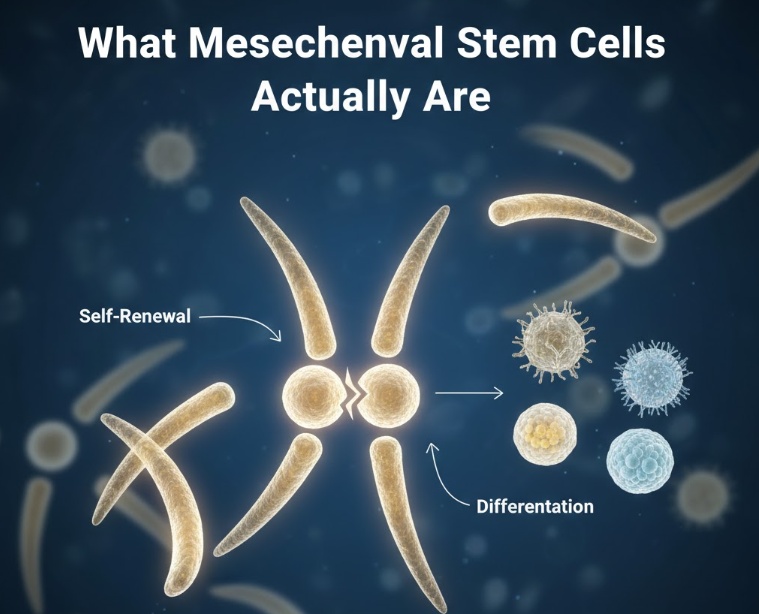
Mesenchymal stem cells are multipotent stromal cells, meaning they are capable of both self-renewal and differentiation into multiple cell types. They were first described by Alexander Friedenstein and colleagues in the 1970s through work on bone marrow stromal cells. Arnold Caplan later coined the term “mesenchymal stem cells” in 1991, and the name stuck even though the scientific community continues to debate exactly how to define and categorize them.
The International Society for Cell and Gene Therapy (ISCT) established a set of minimum criteria for identifying MSCs. To qualify, a cell population must be plastic-adherent when cultured under standard laboratory conditions. It must express the surface markers CD73, CD90, and CD105, while testing negative for CD45, CD34, CD14, and HLA-DR. And it must be capable of differentiating into osteoblasts (bone cells), adipocytes (fat cells), and chondroblasts (cartilage cells) under the right conditions. This trilineage differentiation potential is considered the defining functional test for MSC identity.
Beyond their ability to become other cell types, MSCs serve a number of important biological functions. They support tissue repair and regeneration, regulate immune responses, secrete anti-inflammatory signals, and help maintain the environments where blood-forming cells develop. These functions are primarily carried out through paracrine signaling, which is the process of secreting molecules that influence the behavior of nearby cells. In many therapeutic settings, it is this secretory activity rather than direct differentiation that drives clinical benefit.
MSCs can be derived from a wide range of tissues. Bone marrow was the original source and remains the most studied. Adipose tissue (body fat) became popular because it is abundant and relatively easy to harvest. Other sources include peripheral blood, dental pulp, placenta, amniotic fluid, and umbilical cord tissue. Each source has different biological properties, yields different quantities of cells, and presents different practical challenges. This is where Wharton’s Jelly begins to stand apart.
How Wharton’s Jelly MSCs Are Isolated
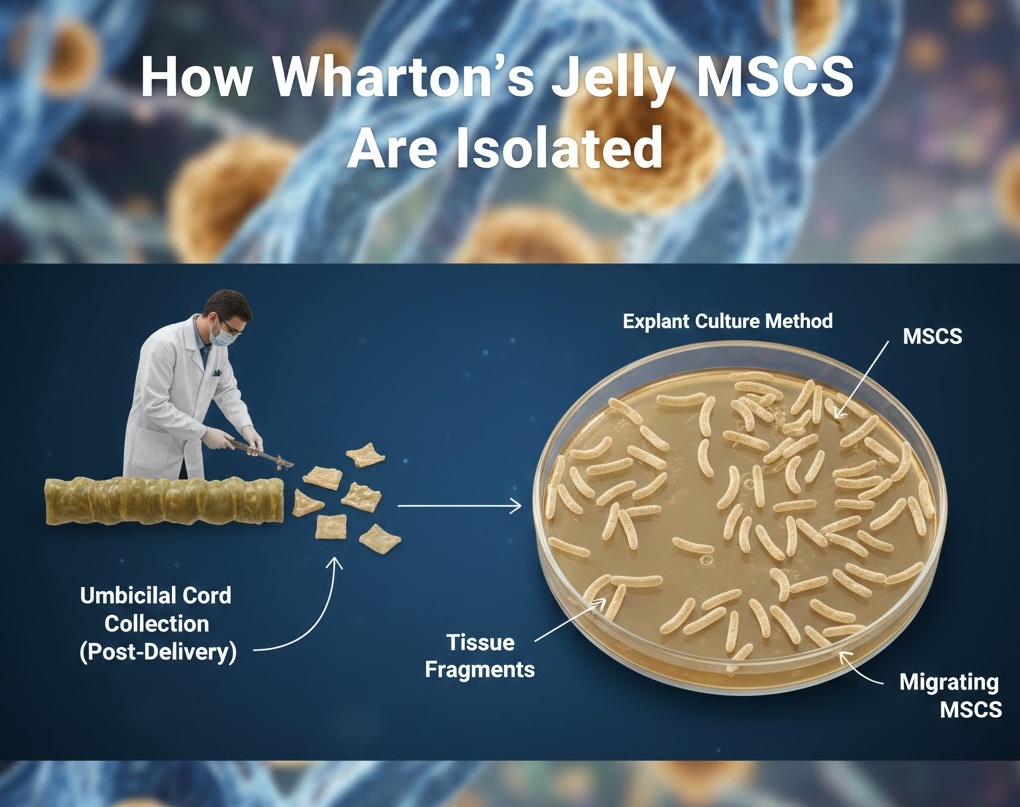
One of the practical advantages of WJ-MSCs is how they are obtained. Umbilical cord collection happens after delivery, with no risk whatsoever to the mother or the newborn. The cord is typically discarded after birth, so collecting it represents no additional medical intervention. With appropriate consent from the parents, the cord is simply preserved and transferred to a processing facility rather than being thrown away.
Once in the lab, researchers use one of two main methods to isolate the MSCs. The explant culture method involves cutting the cord tissue into small fragments and placing them in culture media. Over time, MSCs migrate out from the tissue pieces and begin growing on the culture surface. This method is relatively simple but yields fewer cells per unit of tissue.
The enzymatic digestion method breaks down the extracellular matrix using enzymes such as collagenase, hyaluronidase, and trypsin, releasing the embedded cells into suspension. This approach yields more cells more quickly but requires more processing steps and introduces additional variables that can affect cell quality.
Both methods result in cells that need to be expanded in culture before they can be used clinically. This expansion phase happens under strict GMP (Good Manufacturing Practice) conditions, which are standardized protocols designed to ensure consistency, sterility, and safety. Once expanded to the required quantities, cells are typically cryopreserved, meaning they are frozen and stored for later use.
Before any batch of WJ-MSCs can be used clinically, it goes through quality control testing. This includes flow cytometry to confirm the presence of the right surface markers, sterility testing to rule out contamination, and potency assays to verify that the cells are biologically active. Regulatory bodies including the FDA and the European Medicines Agency have established guidelines for how these products must be manufactured and tested, though the specific regulatory pathways vary depending on how the cells are being used.

Why WJ-MSCs Are Considered the Gold Standard
This is the core question, and the answer is not simple. There is no single property that makes WJ-MSCs superior. Instead, there is a combination of advantages that, taken together, make them a more attractive option than MSCs derived from other sources.
They Produce More Cells
One of the most straightforward advantages is yield. WJ-MSCs can be isolated in significantly higher numbers than bone marrow-derived MSCs, and they proliferate faster in culture. Bone marrow yields are notoriously low, and the quality of bone marrow cells declines sharply with donor age. WJ-MSCs, by contrast, always come from newborns, meaning the donor age variable is essentially eliminated. Research has also shown that WJ-MSCs can sustain more cell divisions before hitting senescence, the state where cells stop dividing and begin losing function. This is important for manufacturing, where you need to expand a small initial sample into millions or billions of cells for clinical use.
Their Immune Profile Is Uniquely Favorable
Perhaps the most clinically significant advantage of WJ-MSCs relates to their interaction with the immune system. All MSCs have some immunomodulatory properties, but WJ-MSCs appear to be particularly potent in this area.
MSCs modulate immune responses through multiple mechanisms. They suppress T-cell proliferation, meaning they can calm down an overactive immune response. They secrete anti-inflammatory cytokines including IL-10, TGF-beta, and prostaglandin E2 (PGE2), as well as an enzyme called indoleamine 2,3-dioxygenase (IDO). WJ-MSCs produce these molecules at higher levels than bone marrow-derived MSCs in most comparative studies.
WJ-MSCs also express lower levels of MHC class I molecules on their surface and virtually no MHC class II molecules. These are the proteins that the immune system uses to identify “self” versus “non-self.” Lower expression of these markers means that WJ-MSCs are less likely to trigger an immune rejection response when transplanted into a patient who is not the original donor. This property, known as low immunogenicity, is what makes allogeneic therapy possible. Allogeneic therapy is when cells from one person are used to treat a different person, essentially creating an off-the-shelf cell therapy product that can be manufactured at scale and stored until needed.
Adding to this picture is the expression of HLA-G, a non-classical MHC molecule that plays a specialized role in immune tolerance. HLA-G is naturally expressed during pregnancy to protect the fetus from the mother’s immune system. WJ-MSCs express HLA-G at relatively high levels, which may contribute to their enhanced immune-tolerogenic properties. This is one of the ways that the fetal origin of these cells translates into a measurable biological advantage.
They Have a More Potent Secretome
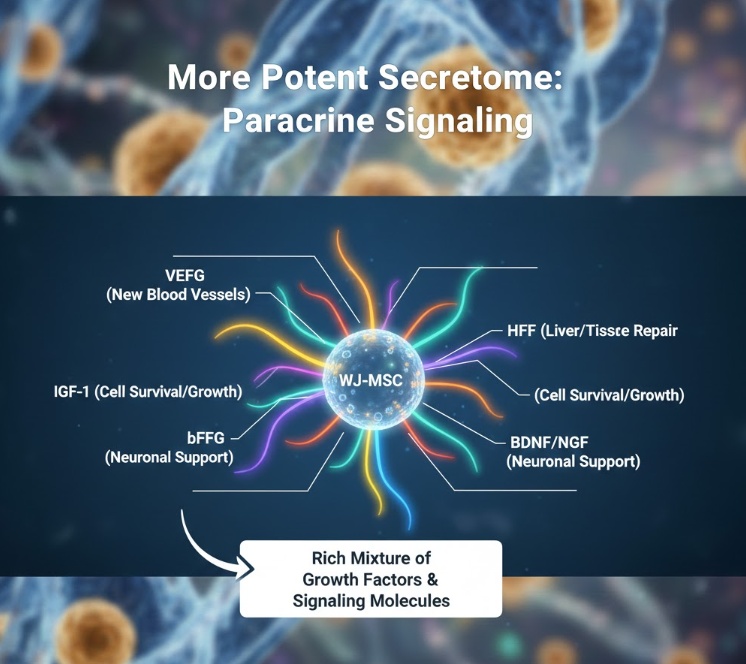
The secretome refers to the full collection of molecules that a cell releases into its surrounding environment. For MSCs, the secretome is arguably more therapeutically important than the cells’ differentiation potential, because most of the healing effects observed in clinical and preclinical settings appear to be driven by paracrine signaling rather than actual cell engraftment or differentiation.
WJ-MSCs secrete a particularly rich mixture of growth factors and signaling molecules. These include vascular endothelial growth factor (VEGF), which promotes the formation of new blood vessels; hepatocyte growth factor (HGF), which supports liver and epithelial tissue repair; insulin-like growth factor 1 (IGF-1), which promotes cell survival and tissue growth; basic fibroblast growth factor (bFGF), which stimulates cell proliferation; and several neurotrophic factors including brain-derived neurotrophic factor (BDNF) and nerve growth factor (NGF), which support the survival and function of neurons.
WJ-MSCs also produce extracellular vesicles (EVs) and exosomes in greater quantities than other MSC sources. These are tiny membrane-bound particles that carry proteins, lipids, and RNA from one cell to another. Research has shown that WJ-MSC-derived exosomes can replicate many of the therapeutic effects of the cells themselves, which has opened up an entire sub-field focused on cell-free therapies using WJ-MSC secretome products.
Their Cells Are Biologically Younger
Age matters enormously in cell biology. As cells age, their telomeres (the protective caps on the ends of chromosomes) shorten, their DNA accumulates damage, they produce more reactive oxygen species (ROS) that can harm surrounding tissue, and they begin expressing proteins associated with cellular senescence. Senescent cells are functionally compromised and can even secrete inflammatory signals that damage nearby tissue.
WJ-MSCs consistently show significantly longer telomeres than bone marrow or adipose-derived MSCs. They produce lower levels of reactive oxygen species. Epigenetic analysis, which looks at patterns of DNA methylation that correlate with biological age, suggests that WJ-MSCs are meaningfully younger than adult-derived MSCs at a molecular level. They also express lower levels of p16 and p21, which are proteins that serve as molecular brakes on cell division and are considered reliable markers of cellular senescence.
What this means practically is that WJ-MSCs are more metabolically active, divide more readily, respond more vigorously to biological signals, and maintain their functional properties through more cell passages in culture. For any therapeutic application where cell potency is critical, this biological youthfulness translates into better outcomes.

Their Differentiation Potential Is Broad
Like all MSCs, WJ-MSCs can differentiate into bone, fat, and cartilage cells under the appropriate conditions. But research suggests they may have a broader differentiation range than MSCs from other sources. Particularly notable is their neurogenic potential. WJ-MSCs appear to be more amenable to neural differentiation than bone marrow-derived MSCs, which has direct implications for neurological disease applications.
Studies have also demonstrated WJ-MSC differentiation toward hepatocyte-like cells (relevant to liver disease), cardiomyocyte-like cells (relevant to heart repair), and endothelial cells (relevant to vascular repair and angiogenesis). This versatility makes WJ-MSCs relevant across a wider range of disease targets than more lineage-restricted stem cell populations.
The Ethical and Practical Advantages Are Significant
Bone marrow harvest requires a surgical procedure performed under general or regional anesthesia, during which a large needle is inserted into the hip bone to extract marrow. It is painful, carries procedural risks, and requires donor recovery time. The quality of the cells harvested declines as the donor ages, meaning that older patients or older healthy donors produce progressively lower quality MSCs.
Embryonic stem cells, while possessing extraordinary differentiation potential, carry significant ethical controversy because their derivation requires the destruction of a human embryo. This has constrained research and clinical translation in many countries.
WJ-MSCs avoid both problems. Collection is non-invasive from any patient’s perspective, causes no harm, requires no recovery, and carries no ethical controversy because it uses tissue that would otherwise be discarded. The fact that the donor is always a newborn means cell quality remains consistent and high regardless of when the cord is collected.
Therapeutic Applications Currently Being Explored
The properties described above have made WJ-MSCs among the most studied cell therapy candidates in the world. Research is ongoing across a broad range of disease areas.
Graft-versus-Host Disease (GvHD) is one of the most well-studied applications. GvHD occurs when immune cells from a bone marrow transplant attack the recipient’s body, and it can be life-threatening. The immunosuppressive properties of MSCs make them a logical treatment candidate, and WJ-MSCs in particular have shown promise in clinical settings.
Autoimmune diseases are a natural fit for any cell therapy with strong immunomodulatory properties. Researchers are investigating WJ-MSC applications in conditions including multiple sclerosis, systemic lupus erythematosus, Crohn’s disease, and rheumatoid arthritis, all of which involve dysregulated immune responses causing tissue damage.
Neurological conditions represent one of the most exciting and challenging areas. The neurogenic differentiation potential of WJ-MSCs, combined with their secretion of neurotrophic factors, has made them candidates for conditions including ALS, Parkinson’s disease, cerebral palsy, and spinal cord injury. The idea that cells injected near or into damaged neural tissue might support survival of existing neurons while potentially differentiating into new neural cells is a compelling one, though significant scientific questions remain about mechanism and delivery.
Cardiovascular repair is another active area. Following a heart attack, the damaged myocardium does not regenerate well on its own. The ability of WJ-MSCs to differentiate toward cardiac-like cells and their secretion of VEGF and other pro-angiogenic factors has made them candidates for post-MI cardiac regeneration protocols.
In orthopedics, researchers are exploring WJ-MSC applications in cartilage repair, bone regeneration, and tendon healing. Cartilage is notoriously difficult to repair naturally because it has limited blood supply and very low regenerative capacity, making it a compelling target for cell-based therapies.
During the COVID-19 pandemic, WJ-MSCs received significant attention as a potential treatment for severe COVID-19 and acute respiratory distress syndrome (ARDS). The reasoning was straightforward: ARDS involves a runaway inflammatory response in the lungs, and WJ-MSCs are potent anti-inflammatory agents. Multiple clinical trials were conducted, and while results were mixed, the episode accelerated research into WJ-MSC manufacturing and delivery.
There are currently hundreds of registered clinical trials on ClinicalTrials.gov involving Wharton’s Jelly or umbilical cord-derived MSCs, covering conditions ranging from autism spectrum disorder to type 2 diabetes to premature aging syndromes.
The Challenges and Limitations Worth Knowing
The scientific case for WJ-MSCs is strong, but it would be misleading to present this field as having all the answers figured out. There are real challenges and open questions that researchers are actively working through.
One of the biggest issues is standardization. There is no universal protocol for isolating, expanding, or characterizing WJ-MSCs. Different labs use different methods, different culture conditions, different passage numbers, and different quality metrics. This makes it extremely difficult to compare results across studies. A product called “WJ-MSCs” from one manufacturer may be meaningfully different from one produced by another lab, and these differences may matter clinically.
Donor-to-donor variability is another complicating factor. Even among newborns, there are differences in gestational age, maternal health, mode of delivery, and other variables that affect the biological properties of the cells. The mechanisms behind this variability are not fully understood, and controlling for it at manufacturing scale remains an active challenge.
The mechanisms by which MSC therapies actually produce their therapeutic effects are also not completely understood. There is strong evidence for paracrine signaling as the dominant mechanism, but the specific molecules responsible for specific effects in specific tissues are not always clear. This “black box” problem makes it harder to rationally design and optimize therapies.
Long-term safety data, particularly around tumorigenicity, remains incomplete. MSCs are not known to be tumorigenic in the way that pluripotent stem cells can be, and no significant tumor formation has been attributed to MSC therapy in published clinical data. But the field is relatively young and long-term follow-up data is still being accumulated.
The translation gap between promising preclinical results and confirmed clinical efficacy is also a genuine concern. Many animal studies have shown dramatic effects from WJ-MSC treatments that have not fully replicated in human trials. This is not unique to MSC research (it is a pervasive problem in biomedical science generally), but it does mean that preclinical results should be interpreted cautiously.
Finally, there is an ongoing scientific debate about whether MSCs are truly “stem cells” in the classical sense of the term. Some researchers argue that the self-renewal and multipotent differentiation potential of MSCs in clinical populations is more limited than the term “stem cell” implies, and that calling them “multipotent stromal cells” or “medicinal signaling cells” (a term Arnold Caplan himself began advocating for) would be more accurate. This terminological debate does not diminish the therapeutic potential of the cells, but it reflects genuine scientific uncertainty about their fundamental biology.
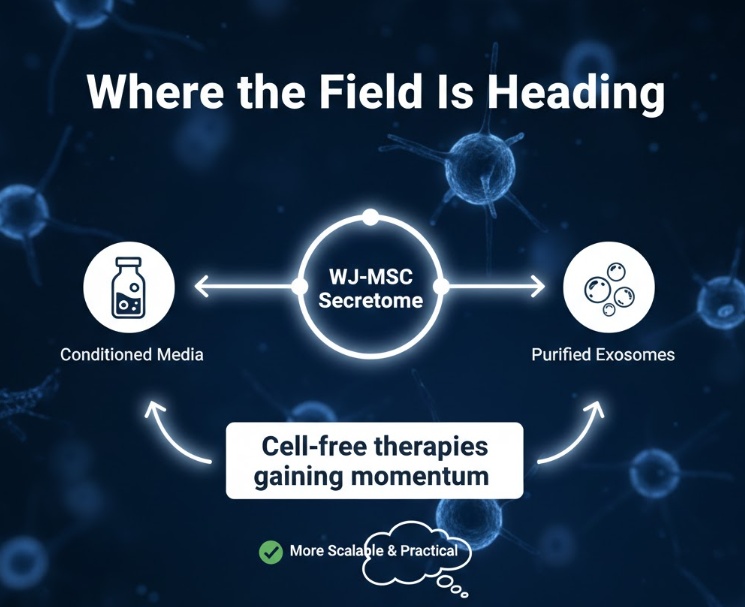
Where the Field Is Heading
Despite these challenges, the direction of WJ-MSC research is clearly forward. Several emerging areas are particularly interesting.
Cell-free therapies based on the WJ-MSC secretome are gaining momentum. Instead of administering live cells (which come with manufacturing complexity, regulatory hurdles, and storage challenges), researchers are exploring whether the conditioned media from WJ-MSC cultures, or purified exosome preparations, can deliver comparable therapeutic effects. Early results are promising, and this approach may eventually be more scalable and practical than live cell therapy.
Genetic engineering of WJ-MSCs is another active area. By introducing genes encoding specific therapeutic proteins or receptors, researchers hope to create enhanced versions of these cells that are better targeted to specific diseases. One approach involves engineering MSCs to express chimeric antigen receptors (CARs), the same technology used in CAR-T cell cancer therapy, adapting it for the MSC context.
Three-dimensional bioprinting using WJ-MSCs as a cellular component is being explored in tissue engineering applications, where the goal is to construct functional tissue structures outside the body for later implantation.
The cord blood and tissue banking industry is expanding rapidly as awareness of WJ-MSC potential grows. Both public and private banking models exist, and the decision about whether to bank cord tissue at birth is becoming more commonly discussed among expecting parents.
The global regenerative medicine market is projected to grow substantially through the end of the decade, with cell therapy products representing a major portion of that growth. WJ-MSCs are positioned as one of the leading candidates for commercial allogeneic cell therapy products, precisely because they can be manufactured at scale, stored, and used off-the-shelf without the need for patient-matched donors.
Why This Matters
The story of Wharton’s Jelly is, at its core, a story about reconsidering something that was assumed to have no value. For centuries, the umbilical cord was seen as nothing more than a biological relay cable, important during pregnancy and useless afterward. The discovery that one of its components contains a population of cells with extraordinary regenerative and immunomodulatory properties is a reminder that medicine regularly finds its most important tools in unexpected places.
The “gold standard” designation for WJ-MSCs is not a marketing slogan. It reflects the weight of comparative evidence showing that these cells are more abundant, more potent, more immunologically compatible, and more practically obtainable than MSCs from other sources. That evidence base is still growing, and the field will continue to evolve as more clinical data becomes available.
What is already clear is that Wharton’s Jelly sits at a rare intersection of biological superiority, ethical simplicity, and manufacturing practicality. That combination does not come along often in medicine. The challenge now is to translate the scientific promise into consistent, well-defined, rigorously tested clinical products that actually help patients. Given how far the field has come in a relatively short period of time, there is good reason to be cautiously optimistic that it will get there.
