Colombia has become one of the more interesting places to watch in regenerative medicine. Over the past decade, clinics in Medellín and Bogotá have quietly built reputations as legitimate destinations for stem cell therapy, drawing patients from across Latin America, the United States, and Europe. And if you look closely at what these clinics are actually offering, a clear pattern emerges: most of them have moved toward allogeneic umbilical cord-derived cells rather than the autologous approaches that were more common a generation ago.
To understand why, you need to understand what separates these two methods in the first place, what makes cord cells scientifically interesting, and how Colombia’s specific circumstances have made this shift both practical and logical.
The Difference Between Autologous and Allogeneic Therapy
Stem cell therapy is not one thing. The term covers a wide range of treatments that differ significantly depending on where the cells come from and how they are used.
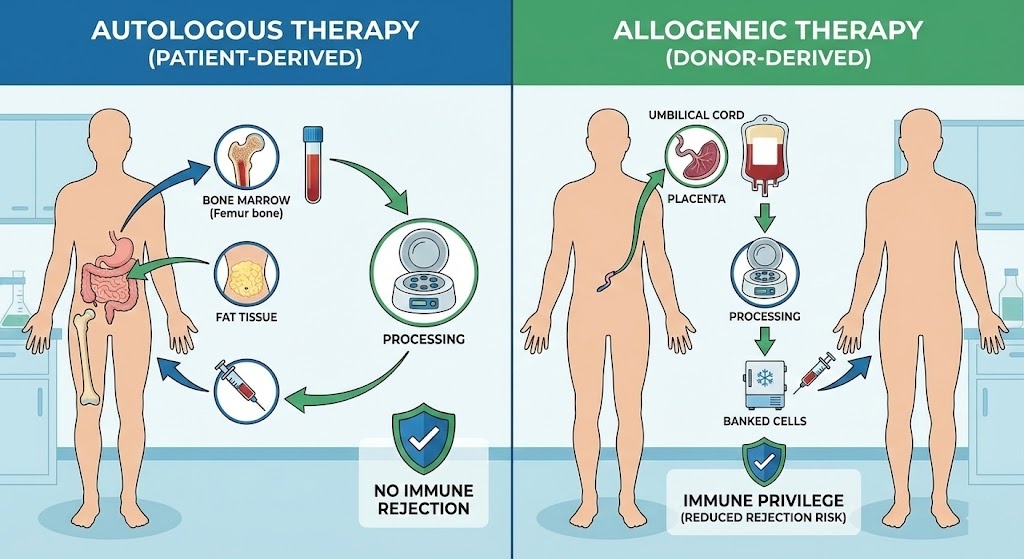
Autologous therapy means the cells come from the patient receiving the treatment. A doctor harvests cells from the patient’s own body, typically from bone marrow, fat tissue (adipose), or peripheral blood, processes them in a lab, and then reintroduces them into the same patient. Because the cells are the patient’s own, there is no immune mismatch to worry about. The body recognizes the cells as self and does not reject them. This approach has a long history in oncology, particularly in bone marrow transplants following chemotherapy, and has expanded into orthopedic and autoimmune applications over time.
Allogeneic therapy works differently. Here, the cells come from a donor. They are collected, screened, processed, and banked, then used in patients who have no biological connection to the original donor. The cells are, essentially, an off-the-shelf product. The obvious concern with this approach is immune rejection, which is a real issue with some types of donor transplants. But certain cell types, particularly mesenchymal stem cells derived from umbilical cord tissue, have properties that make immune rejection far less of a problem than you might expect.
What Makes Umbilical Cord Cells Different
Not all stem cells behave the same way. The source of a cell matters enormously, and umbilical cord-derived cells have several properties that make them stand out from other options.
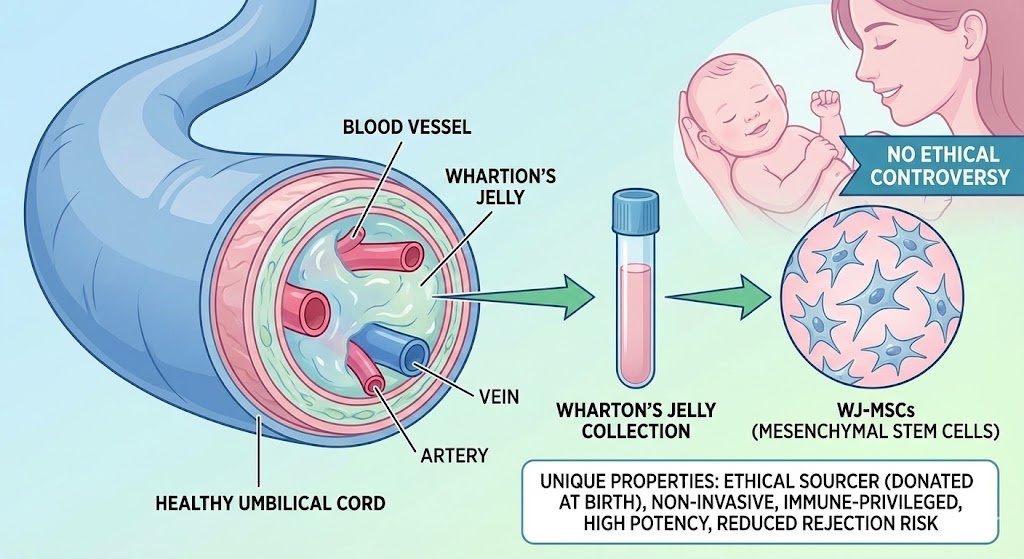
Umbilical cord tissue is collected at birth from donated cords that would otherwise be discarded. There is no ethical controversy involved and no invasive procedure required from the donor. The cells collected from cord tissue, particularly from a gelatinous substance inside the cord called Wharton’s jelly, are classified as mesenchymal stem cells (MSCs). These Wharton’s jelly-derived MSCs, often abbreviated as WJ-MSCs, have become the preferred cell type for many regenerative applications precisely because of how they behave once introduced into a patient.
One of the most important characteristics of WJ-MSCs is their immune profile. These cells express very low levels of the surface markers that typically trigger an immune response in a recipient. In practical terms, this means the body is far less likely to recognize and attack them compared to other types of donor tissue. This low immunogenicity is part of why allogeneic cord cell therapy can work across unrelated individuals without the immune-suppressing drugs normally required for transplants.
Beyond their immune properties, cord cells also have a significant potency advantage over cells collected from adult donors. Stem cells age along with the rest of the body. A 65-year-old patient who undergoes an autologous harvest will receive cells that reflect decades of oxidative stress, inflammation, and biological wear. The regenerative signaling capacity of those cells is measurably lower than what you would find in cells collected from a newborn cord. For older patients or those with chronic illness, this difference is not trivial. Umbilical cord cells are consistently young, consistently potent, and relatively predictable in quality because of how they are collected and processed.
Cord blood, which contains a different type of stem cell called hematopoietic stem cells (HSCs), is also collected from umbilical cords but is used primarily in blood and immune system disorders. When Colombian clinics and regenerative medicine providers talk about cord cells for conditions like osteoarthritis, autoimmune diseases, or neurological conditions, they are usually referring to WJ-MSCs from cord tissue rather than cord blood. The distinction matters clinically, even though both come from the same source.
Colombia’s Regulatory Environment
One reason Colombian clinics have been able to move aggressively toward allogeneic cord therapies is that the country’s regulatory framework allows for more flexibility than what you would find in the United States or the European Union.
In the U.S., the FDA’s oversight of stem cell products has tightened considerably over the past several years. Treatments that do not fall under specific exemptions face a lengthy and expensive approval process, and many clinics offering stem cell therapies have faced enforcement action. The EU has similarly strict controls through the European Medicines Agency. These regulations exist for good reasons, but they also slow adoption and limit patient access within those markets.
Colombia’s regulatory body, INVIMA (Instituto Nacional de Vigilancia de Medicamentos y Alimentos), has been developing its framework for advanced therapy medicinal products with an approach that balances safety oversight with room for clinical application. Provisions for compassionate use and hospital exemption protocols give Colombian clinics pathways to administer therapies that would face much steeper barriers elsewhere. This does not mean anything goes in Colombia, but it does mean that clinics operating within the proper institutional structures have meaningful access to allogeneic cord cell products without the years-long bottleneck that would exist in other regulatory environments.
This flexibility has attracted serious investment and clinical development, not just fringe operators. The clinics that have built genuine reputations in this space are working within Colombia’s legal and institutional frameworks, not around them.
The Practical Case for Allogeneic Cord Cells
Beyond the science and the regulations, there are straightforward logistical reasons why allogeneic cord cells work better for clinical operations than autologous approaches.
Autologous therapy requires a surgical harvest. A patient needs to come in, undergo a procedure to extract bone marrow or fat tissue, wait for the lab to process the cells, and then return for the infusion. That entire sequence can take days or weeks, and it introduces multiple points where something can go wrong. The harvest itself carries procedural risks. The processing requires patient-specific lab work. If the patient is traveling from another country for treatment, the timeline becomes even more complicated.
Allogeneic cord products do not have any of those constraints. The cells are already processed, banked, and quality-tested before a patient ever walks through the door. A clinic can screen a patient, confirm eligibility, and administer the treatment on a much shorter timeline. For a Colombian clinic serving medical tourists from the United States or elsewhere in Latin America, this off-the-shelf availability is a significant operational advantage.
Cost follows from this as well. Patient-specific processing is expensive. When a lab has to handle each patient’s cells as a unique batch, that labor and infrastructure cost gets passed along. Allogeneic products can be manufactured at scale, which brings the per-unit cost down while allowing the same quality standards to be applied consistently. Colombian clinics can therefore offer treatments at price points that are genuinely competitive on a global basis, which is part of what drives medical tourism to the country in the first place.
Who Is Seeking These Treatments
The patient population coming to Colombian clinics for stem cell therapy also shapes why allogeneic cord cells make more sense in this context than autologous approaches.
A significant portion of patients seeking regenerative therapy are middle-aged to elderly. They are dealing with conditions like osteoarthritis, autoimmune disorders, neurological decline, or metabolic disease. These are the exact patients for whom autologous cell quality is most likely to be compromised. An 70-year-old with advanced osteoarthritis who undergoes a bone marrow harvest is not going to get the same regenerative output from those cells that a younger, healthier person would. The cells are simply older, and their capacity to reduce inflammation and support tissue repair reflects that age.
International patients, especially those coming from the U.S. and Europe where similar treatments are either unavailable or prohibitively expensive, represent a large share of the demand. These patients are often well-informed. They have done research, consulted with physicians at home, and chosen to come to Colombia after comparing their options. For this group, standardized allogeneic products are often preferable because the quality control is more predictable. When a cell product has been screened, processed under documented protocols, and banked with traceability records, there is less variability than in a patient-specific harvest whose outcome depends heavily on the individual’s own biology.
Colombia’s domestic patient population is also relevant here. The country has a significant and growing aging population, along with high rates of conditions that have driven interest in regenerative approaches. Local demand, combined with international medical tourism, creates the volume that makes investment in quality cord cell infrastructure economically viable.
What the Clinical Evidence Shows
It is worth being clear about where the science actually stands, because this is an area where clinic marketing and peer-reviewed research do not always tell the same story.
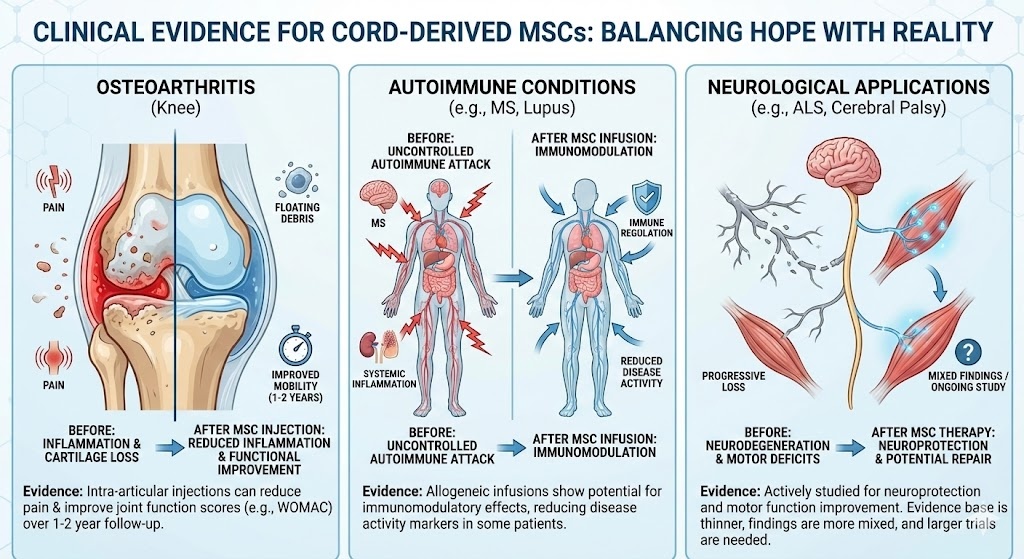
There is genuine published evidence supporting the use of MSCs, including cord-derived MSCs, in a range of conditions. Studies in osteoarthritis have shown that intra-articular injection of MSCs can reduce pain and improve function over follow-up periods of one to two years. Research in autoimmune conditions, particularly multiple sclerosis and systemic lupus, has shown that allogeneic MSC infusions can produce immunomodulatory effects that reduce disease activity in some patients. Neurological applications, including conditions like ALS and cerebral palsy, are also being actively studied, though the evidence base here is thinner and the findings more mixed.
What the evidence does not support, at least not yet, is the broad range of near-miraculous claims that appear on some clinic websites. The honest position is that cord cell therapy is a genuinely promising area of medicine that has demonstrated real effects in certain applications, while remaining insufficiently studied in others. Patients evaluating treatment options deserve that honest framing rather than overpromised outcomes.
Colombian clinics with serious research affiliations and transparent protocols tend to be more measured in their claims. They are also more likely to be the ones actually contributing to the evidence base through institutional trial work and outcome tracking.
When Autologous Therapy Still Makes Sense
The shift toward allogeneic cord cells in Colombia does not mean autologous therapy has no place in modern regenerative medicine. There are situations where using a patient’s own cells remains the clinically appropriate choice.
Certain oncology applications, particularly in hematology, require precisely matched immune profiles that autologous approaches naturally provide. For a patient whose own cells are needed to reconstitute a destroyed immune system after aggressive cancer treatment, the match is the point. No allogeneic product can replicate that.
Some patients also have a strong personal preference for using their own biological material, and in younger, healthier individuals with good cell quality, autologous harvests can produce perfectly viable treatments. For a 35-year-old with a sports-related joint injury, an autologous PRP or bone marrow concentrate procedure may be entirely appropriate and cost-effective.
The key is that neither approach is universally superior. The best choice depends on the patient’s age and health status, the condition being treated, the quality of available allogeneic products, and the clinical judgment of the treating physician.
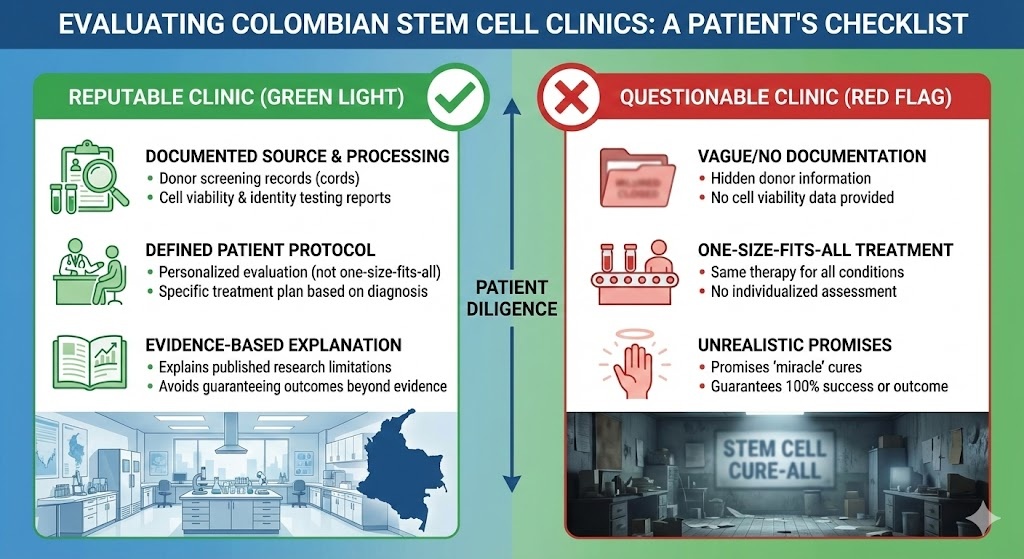
How to Evaluate a Colombian Stem Cell Clinic
Because Colombia’s regulatory flexibility is one of the draws for both clinics and patients, it also creates space for providers operating at very different standards. Not every clinic offering stem cell therapy in Medellín or Bogotá is doing so with the same level of scientific rigor, quality control, or institutional oversight.
Patients considering treatment should look for clinics that can provide documentation on the source and processing of their cord cell products, including donor screening records and cell viability testing. Reputable providers will have a defined protocol for patient evaluation and will not offer the same treatment to every patient regardless of diagnosis. They should be willing to explain what the evidence supports for a given condition and what it does not, rather than promising outcomes that go beyond what published research has shown.
Institutional affiliations, whether with Colombian universities, hospitals, or research bodies, are a useful signal. So is membership in organizations that hold practitioners to published standards in regenerative medicine. The absence of these markers does not automatically indicate a bad actor, but their presence provides meaningful accountability.
Where This Is All Heading
Colombia is not a finished story in regenerative medicine. The country is still developing the infrastructure, research output, and regulatory clarity that will define its long-term position. But the trajectory is toward a more formalized, research-integrated industry rather than away from it.
Investment in cord blood and tissue banking has been growing in major Colombian cities. Research partnerships between Colombian clinical institutions and international universities are producing publications that add to the global evidence base. And as the market matures, the gap between serious clinical providers and wellness-oriented operations is likely to widen, making it easier for patients to identify where the real medicine is happening.
The preference for allogeneic umbilical cord cells that is visible today in Colombian clinics is not a fad. It reflects a convergence of good science, practical logistics, patient demographics, and a regulatory environment that allows innovation to move faster than it can in more heavily restricted markets. Whether the rest of the world catches up to this model or Colombia continues to lead it, the cells themselves are likely to remain central to how regenerative medicine develops in the region.
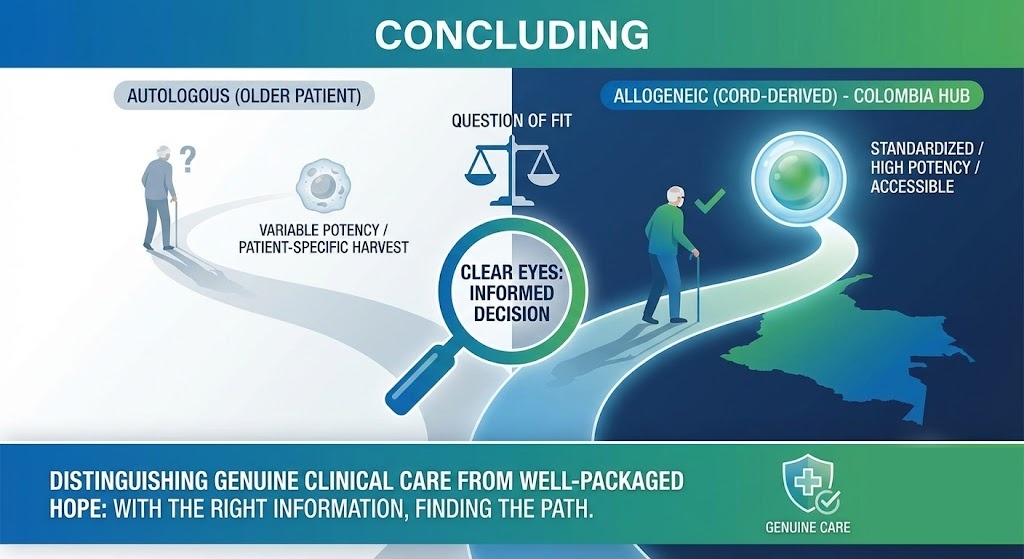
Final Thoughts
The autologous versus allogeneic debate is not really a debate. It is a question of fit. For a growing share of patients, particularly older patients with chronic conditions seeking treatment in Colombia, allogeneic umbilical cord-derived cells offer a combination of potency, standardization, and accessibility that patient-specific harvests simply cannot match. Colombian clinics have recognized this, and their move toward cord cell therapy reflects both the science and the practical realities of operating in that specific market.
For anyone exploring regenerative medicine options, whether locally or as a medical tourist, the most important thing is to approach the topic with clear eyes. The potential is real. So is the need to distinguish genuine clinical care from well-packaged hope. The good news is that in Colombia, both exist, and with the right information, one is not that hard to find.