Your body is doing something remarkable right now, without you thinking about it for even a second. Deep inside your bones, billions of blood cells are dying off and being replaced. The lining of your gut renews itself every few days. Skin cells flake away and new ones push up to take their place. This constant, quiet repair work is made possible by one of the most important types of cells in the human body: stem cells.
For decades, stem cells have sat at the center of some of the most exciting and controversial conversations in science. They have been heralded as the key to treating diseases once considered untreatable, and they have also been at the heart of serious ethical debates. They have attracted billions of dollars in research funding, drawn in Nobel Prize winners, and unfortunately also attracted unscrupulous clinics willing to exploit desperate patients.
So what exactly are stem cells? How do they work? And what does the field of regenerative medicine actually look like beyond the headlines? This article walks through all of it, from the basic biology to the clinical realities, in a way that anyone willing to pay attention can follow.
What Are Stem Cells?
To understand stem cells, it helps to start with what makes them different from the other 200-plus cell types in the human body.
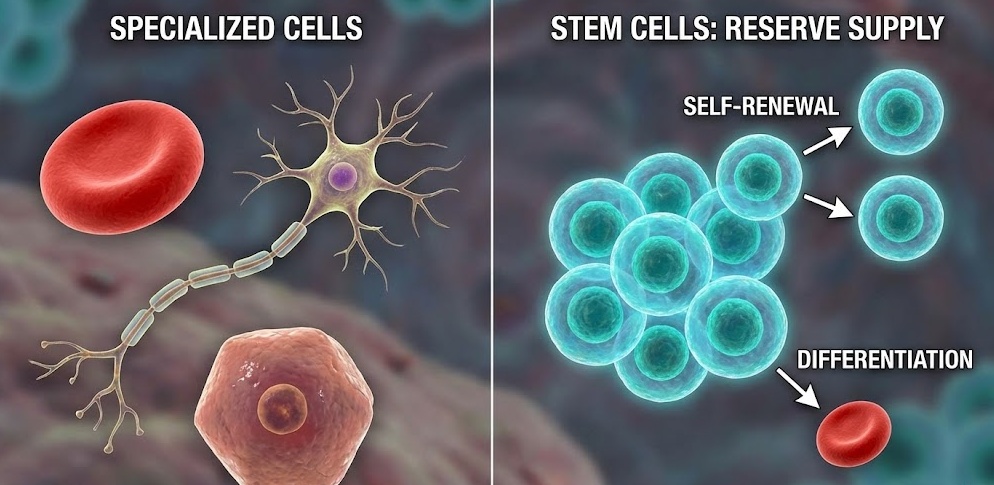
Most cells in your body are specialists. A liver cell does liver things. A neuron fires electrical signals. A red blood cell carries oxygen. These cells are fully committed to their jobs, and they cannot become something else. They also have limited ability to reproduce themselves. A mature red blood cell, for example, cannot divide at all.
Stem cells work differently in two important ways. First, they can divide and produce copies of themselves, sometimes for very long periods of time. This is called self-renewal. Second, they have the ability to develop into more specialized cell types. That process is called differentiation.
Together, these two abilities make stem cells the body’s reserve supply of raw material. When tissue gets damaged or worn out, stem cells can activate, divide, and send daughter cells off to become whatever the body needs in that location.
The Potency Spectrum
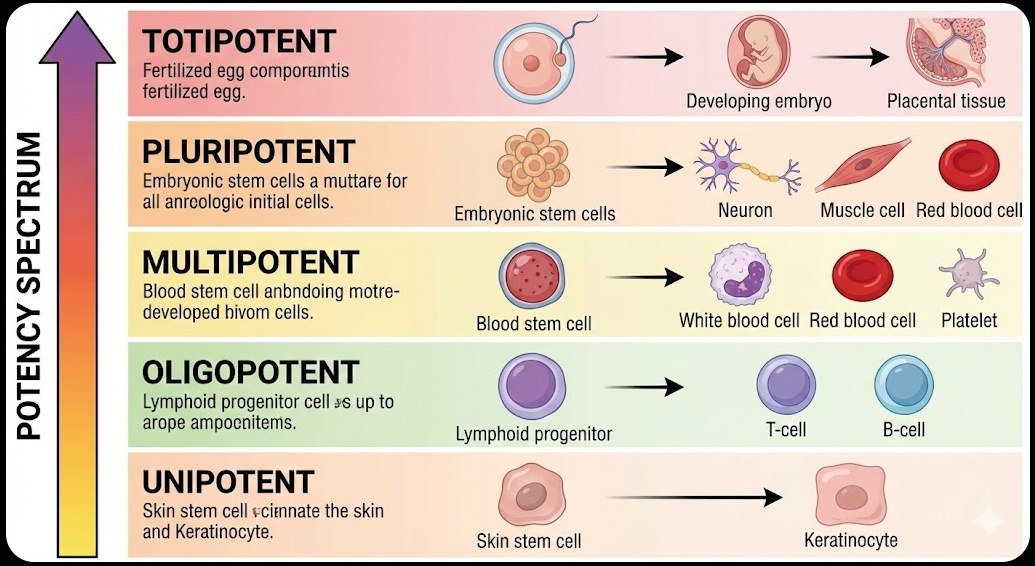
Not all stem cells are created equal. Scientists use the term “potency” to describe how many different cell types a stem cell can potentially become, and there is a wide range.
At the top of the spectrum sit totipotent cells. A fertilized egg is totipotent, meaning it has the ability to develop into every single cell type needed for a complete organism, including the cells that make up the placenta. This level of flexibility lasts only for the first couple of cell divisions after fertilization.
Just below totipotent are pluripotent stem cells. These can become virtually any cell type in the body, but they can no longer form placental tissue. Embryonic stem cells fall into this category, which is one reason they generate so much scientific interest.
Multipotent stem cells are one step more restricted. They can still differentiate into several cell types, but only within a certain category. Blood stem cells, for example, are multipotent: they can become red blood cells, white blood cells, and platelets, but they cannot become neurons or skin cells.
Further down the spectrum are oligopotent cells, which are limited to just a few closely related cell types, and unipotent cells, which can only produce one specific cell type. Skin stem cells are a good example of the latter: they keep producing new skin cells, and that is essentially their whole job.
Where Do Stem Cells Come From?
Stem cells are found in several places, and their source matters quite a bit for both research and therapy.
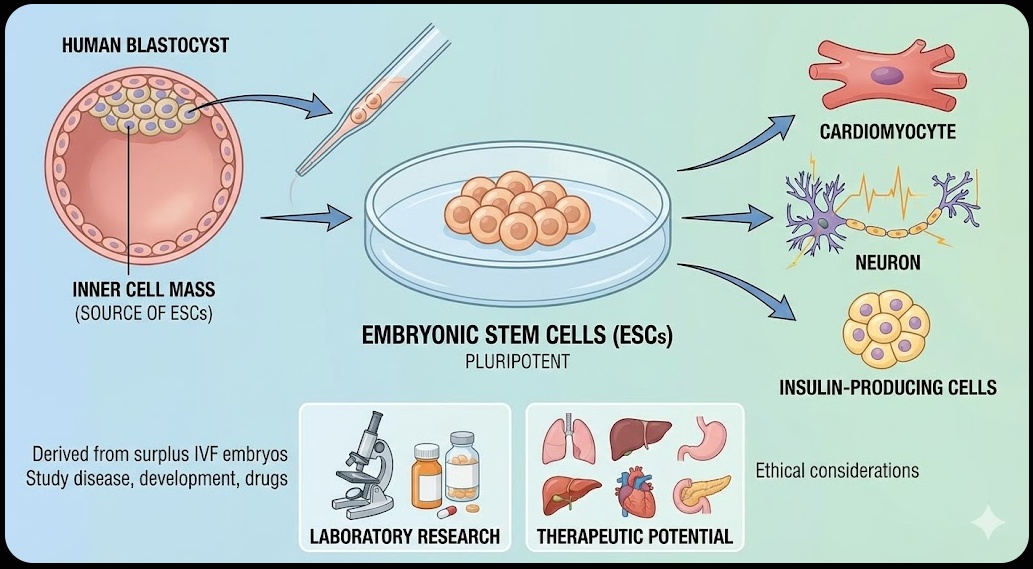
Embryonic stem cells come from embryos at a very early stage of development called the blastocyst, which forms about five days after fertilization. At this point, the embryo is a hollow ball of around 150 cells, and the inner cell mass of that ball contains pluripotent stem cells. These can be removed and grown in lab conditions indefinitely.
Adult stem cells, sometimes called somatic stem cells, exist throughout the body in small populations. They have been found in bone marrow, fat tissue, the brain, the liver, skin, and the lining of the gut, among other places. Their primary job is maintenance: replacing worn-out cells in their home tissue.
Umbilical cord blood is a third source. After a baby is born, the blood remaining in the umbilical cord and placenta is rich in blood-forming stem cells. This material is sometimes collected and stored in cord blood banks.
Induced pluripotent stem cells, commonly called iPSCs, are a more recent and particularly significant category. These are ordinary adult cells, like skin or blood cells, that have been genetically reprogrammed back into a pluripotent state in the laboratory. They behave a great deal like embryonic stem cells but without requiring an embryo.
Finally, perinatal stem cells can be harvested from amniotic fluid and placental tissue, providing another source that avoids many of the controversies tied to embryonic material.
The Major Types of Stem Cells, Explained
Embryonic Stem Cells
Embryonic stem cells (ESCs) were first successfully isolated from human embryos in 1998, and the discovery changed the trajectory of biological research. Because they are pluripotent, they have the theoretical ability to be coaxed into becoming any cell type needed for therapy: heart muscle cells, neurons, insulin-producing beta cells, and so on.
In the lab, ESCs have proven enormously useful for studying how early human development works, how diseases begin at the cellular level, and how drugs affect developing cells. They have served as a kind of benchmark for what pluripotency looks like, and they remain important research tools.
However, their use comes with a significant ethical problem that has not gone away. Harvesting embryonic stem cells destroys the embryo they come from. For researchers and medical institutions in many countries, this is acceptable because the embryos used are typically surplus embryos from in vitro fertilization clinics that would otherwise be discarded. For others, including many religious groups and some ethicists, destroying a human embryo at any stage crosses a moral line.
Regulations around ESC research vary sharply from one country to the next. Some countries have few restrictions. Others have partial restrictions tied to the source of the embryo. A few have banned the practice entirely. In the United States, federal funding for new embryonic stem cell line creation has faced various restrictions over the years, though private funding has continued.
Adult Stem Cells
Adult stem cells are far less flexible than embryonic ones, but they come with a much simpler ethical profile and have already proven their clinical usefulness. In fact, the most widely used stem cell therapy in the world, the bone marrow transplant, relies entirely on adult stem cells.
Among adult stem cells, two varieties have received particular attention.
Hematopoietic stem cells (HSCs) live in bone marrow and are responsible for generating all the different types of blood cells throughout a person’s life. They are well-understood, relatively accessible, and have been used in transplants to treat blood cancers and certain inherited blood disorders for more than 60 years.
Mesenchymal stem cells (MSCs) are found in bone marrow, fat tissue, and various other locations. They can differentiate into bone, cartilage, muscle, and fat cells, which makes them interesting candidates for repairing musculoskeletal damage. They also have anti-inflammatory properties that researchers believe may be useful in treating a range of conditions. MSCs are among the most studied and most frequently tested stem cell types in clinical trials today.
The main limitation of adult stem cells is their restricted potency. Getting them to reliably differentiate into cell types outside their normal range is difficult, and they are generally harder to grow in large quantities than embryonic stem cells.
Induced Pluripotent Stem Cells
In 2006, a Japanese scientist named Shinya Yamanaka published a paper that genuinely surprised the scientific community. His lab had figured out how to take mature skin cells from mice and reprogram them back into a pluripotent state by introducing just four specific genes. The following year, his team and another group led by James Thomson independently replicated the feat using human cells.
Yamanaka received the Nobel Prize in Physiology or Medicine in 2012, a recognition of just how significant the discovery was.
The cells produced by this reprogramming process, called induced pluripotent stem cells, share many of the key properties of embryonic stem cells. They can divide indefinitely and can be guided to differentiate into a wide variety of cell types. But because they can be made from a patient’s own cells, they offer two important advantages: they sidestep the ethical issues around embryo use, and they could theoretically be used to produce personalized therapies that the patient’s immune system would not reject.
iPSCs are not without their own problems. The reprogramming process is still relatively inefficient, meaning many attempts are needed to produce a small number of viable cells. There are also concerns about genomic instability, where errors introduced during reprogramming can potentially lead to abnormal cell growth or even cancer. Researchers are actively working on safer and more efficient reprogramming methods.
Cord Blood and Perinatal Stem Cells
Cord blood banking has grown into a significant industry over the past two decades. Parents are often offered the option of storing their newborn’s cord blood at private facilities for a recurring fee, with the idea that the stem cells inside could potentially be used later in life if the child develops a blood disorder or certain other conditions.
The stem cells found in cord blood are primarily hematopoietic, meaning they are best suited to treating blood-related diseases, much like bone marrow transplants. Cord blood transplants have been performed successfully for conditions like leukemia, sickle cell disease, and certain immune deficiencies.
Researchers are also exploring stem cells found in amniotic fluid and placental tissue, which can be collected during routine prenatal procedures or at birth without harm to the mother or baby. These cells show some multipotent properties and are being studied for potential use in treating various conditions, though they have not yet entered widespread clinical use.
How Stem Cells Actually Work: The Biology
Understanding stem cells is not just about knowing what they are. The more interesting question is how they know what to do.
Self-Renewal and Division
Stem cells divide in one of two ways. Symmetric division produces two identical daughter cells, both of which remain stem cells. This allows a stem cell population to expand. Asymmetric division produces one daughter cell that remains a stem cell and one that begins the process of differentiating into a specialized cell. This is how the stem cell pool maintains itself while also supplying the body with new specialized cells.
The environment surrounding a stem cell, called the niche, plays a major role in regulating this behavior. The niche is made up of neighboring cells, the proteins and molecules that fill the space between cells (the extracellular matrix), and the chemical signals floating through the tissue. Together, these factors send instructions to the stem cell: stay in a resting state, divide, or differentiate.
Several signaling pathways are particularly important in stem cell regulation. The Wnt pathway helps control self-renewal in many stem cell types, including those in the gut and bone marrow. The Notch pathway influences whether stem cells stay undifferentiated or begin to specialize. The Hedgehog pathway plays a role in tissue development and repair. When these pathways are disrupted by disease or mutation, the consequences can range from impaired healing to the development of cancer.
Differentiation: How a Stem Cell Becomes Something Specific
When a stem cell receives the right combination of signals, it begins the process of becoming a specialized cell. This is differentiation, and it happens in steps rather than all at once.
The signals that trigger differentiation can come from growth factors, hormones, or direct contact with neighboring cells. These signals activate or silence specific genes, which in turn changes which proteins the cell produces. As different genes are turned on and off, the cell begins to look and behave differently.
Transcription factors are proteins that act like switches for gene expression, and they are central to differentiation. Certain transcription factors are associated with specific cell fates: particular combinations push a cell toward becoming a neuron, a muscle cell, a liver cell, and so on.
Epigenetic changes add another layer to this process. Epigenetics refers to modifications that affect how genes are read without actually changing the DNA sequence. Chemical tags called methyl groups can be added to DNA to silence genes, and proteins called histones that help package DNA can be modified to make certain genes more or less accessible. During differentiation, these epigenetic changes help lock a cell into its new identity.
More Than Replacement: Paracrine Signaling
One of the more surprising things researchers have discovered about stem cells is that their therapeutic effects are not always about actually replacing damaged tissue. In many cases, stem cells influence healing by releasing signaling molecules into their surroundings.
This process, called paracrine signaling, involves the secretion of growth factors, cytokines, and other compounds that tell nearby cells to repair themselves, reduce inflammation, form new blood vessels, or resist cell death. Stem cells also release tiny membrane-enclosed particles called extracellular vesicles, which carry signals and can influence cells at a distance.
This finding has opened up a parallel line of research: so-called cell-free therapies that use the molecules stem cells produce rather than the cells themselves. If the beneficial effects of stem cells come largely from their secretions, then it may eventually be possible to deliver those benefits without the complications of transplanting live cells.
Stem Cells in Regenerative Medicine
Regenerative medicine is the field of medicine focused on repairing, replacing, or regenerating tissues and organs that have been damaged by disease, injury, or aging. It draws on three main approaches: cell therapy (putting new cells into the body), tissue engineering (building tissues and organs in the laboratory), and gene therapy (correcting faulty genes). Stem cells are relevant to all three.
Treatments That Are Already Established
The most established stem cell therapy by far is the bone marrow transplant, which has been performed since the late 1960s. In this procedure, a patient’s diseased bone marrow is destroyed, usually with chemotherapy, radiation, or both, and then replaced with healthy hematopoietic stem cells from a donor or from the patient’s own previously collected cells. This approach has saved countless lives in patients with leukemia, lymphoma, aplastic anemia, and certain inherited immune deficiencies.
Stem cells have also been used in treating burns for decades. Researchers learned to take small samples of a burn patient’s own skin, cultivate the stem cells within that tissue in a lab, and then grow sheets of new skin that could be grafted onto the wound. This technique has significantly improved survival rates in patients with severe burns covering large areas of the body.
Another established application involves the cornea. The cornea relies on a ring of stem cells at its edge, called limbal stem cells, to maintain its surface. When these cells are damaged or destroyed by injury or chemical burns, the surface of the eye becomes progressively scarred and vision deteriorates. Transplanting healthy limbal stem cells from a donor eye or even from the patient’s unaffected eye has been shown to restore the corneal surface and recover significant vision in many patients.
Blood disorders like sickle cell disease and thalassemia have also been treated with stem cell transplants, again primarily through the bone marrow route.
Where Research Is Headed: Emerging Applications
Beyond what is already in clinical use, stem cell research is targeting a remarkable range of conditions.
Neurological Conditions
The brain and spinal cord have very limited ability to repair themselves after injury, which is why conditions like Parkinson’s disease, ALS, and spinal cord injury can be so devastating. Stem cell research in this area is trying to solve two different problems at once: replacing lost neurons and protective cells, and creating an environment where any remaining healthy cells can survive and function.
In Parkinson’s disease, the specific neurons that die are dopamine-producing cells in a region of the brain called the substantia nigra. Clinical trials are now testing whether pluripotent stem cell-derived dopamine neurons can be transplanted into patients and survive well enough to reduce symptoms. Early results have been cautiously promising, though the work is still in early phases.
For spinal cord injury, researchers are testing the use of stem cell-derived oligodendrocyte progenitor cells, which help rebuild the myelin sheath that insulates nerve fibers. Trials in this area have been going on for several years with results suggesting at least some potential for improving function in recently injured patients.
Cardiovascular Disease
When the heart muscle is damaged by a heart attack, the lost cells are replaced largely by scar tissue, which cannot contract and pump blood the way normal heart muscle does. This is a major reason why heart failure often follows a significant heart attack.
Researchers have been testing various stem cell types, particularly mesenchymal stem cells and cardiac progenitor cells, to see whether introducing them into damaged heart tissue can help restore function. Results across different trials have been mixed. Some showed modest improvements in heart function, while others showed little effect. The field is currently trying to identify which patient populations benefit most, which cell types work best, and how delivery method affects outcomes.
Diabetes
Type 1 diabetes results from the immune system attacking and destroying the beta cells in the pancreas that produce insulin. Replacing those beta cells has been a long-standing goal in diabetes research.
Scientists have now successfully coaxed human pluripotent stem cells, including iPSCs, into becoming insulin-producing beta-like cells in the laboratory. Several companies and research institutions are running or planning clinical trials to test whether these lab-grown cells can be transplanted into patients and restore blood sugar regulation. This is considered one of the more near-term realistic goals in stem cell therapy.
Orthopedic Applications
Cartilage is notoriously difficult to repair because it has essentially no blood supply and very limited regenerative capacity on its own. This is why osteoarthritis, which involves the gradual breakdown of cartilage in joints, is such a persistent and painful problem.
Mesenchymal stem cells have shown some ability to differentiate into cartilage-producing cells called chondrocytes, and there is ongoing research into whether injecting these cells into damaged joints or using them to grow replacement cartilage tissue in the lab can provide long-term relief. Several approaches are being tested in clinical trials, with early results suggesting modest improvements in pain and function for some patients.
Other Promising Areas
Research is also underway into using stem cells to treat liver failure by generating functional liver cells, autoimmune diseases like multiple sclerosis and Crohn’s disease by resetting the immune system, and vision loss from conditions like age-related macular degeneration by replacing the retinal pigment epithelial cells that die in the course of that disease. None of these approaches have yet become standard treatments, but several are in clinical trials.
Tissue Engineering and Organoids
One of the most visually striking developments in stem cell science is the use of these cells to grow tissue structures outside the body.
In tissue engineering, stem cells are seeded onto a scaffold made from natural or synthetic materials and then provided with the right signals to differentiate and organize themselves into functional tissue. Researchers have grown small patches of heart muscle, cartilage, and even functional human skin using this approach. Some of these engineered tissues have already been used in patients.
Organoids represent a related but distinct development. These are small, three-dimensional tissue structures grown entirely in the lab from stem cells, without a scaffold, that self-organize into miniature versions of organs. Scientists have grown brain organoids, intestinal organoids, kidney organoids, and more. These structures are not functional organs in any complete sense, but they are remarkably useful for studying how organs develop, how diseases affect them, and how drugs interact with human tissue in ways that animal models often cannot capture.
The long-term vision of the field is the production of complete functional organs for transplant, grown from a patient’s own cells and therefore unlikely to be rejected. That goal remains far off, but the progress in tissue engineering has been steady.
How Stem Cell Therapies Are Delivered
Getting stem cells into a patient’s body in a way that they survive, reach the right location, and do what they are supposed to do is one of the practical challenges the field grapples with constantly.
Autologous vs. Allogeneic Transplants
An autologous transplant uses cells taken from the patient who will receive them. The advantage is that immune rejection is not an issue: the cells are recognized as self. The disadvantage is that the process takes time, and if the patient is very ill, their own cells may already be compromised in some way.
An allogeneic transplant uses cells from a donor. These are more immediately available and can potentially be produced in quantity in advance, but they carry the risk of immune rejection. The donor’s immune cells can also attack the recipient’s body, a complication called graft-versus-host disease, or GvHD, which can range from mild and manageable to life-threatening.
iPSC technology raises the prospect of creating large batches of “universal donor” cells, engineered to evade immune rejection, that could be used off the shelf for any patient without the need for matching.
Methods of Delivery
The method used to get stem cells into the body depends on the condition being treated and the target tissue.
Intravenous infusion is the simplest approach and is used for blood and immune system conditions, where the cells naturally home to bone marrow. Direct injection into a specific tissue or organ is used when cells need to reach a localized area, such as the heart muscle or the brain. Surgical implantation, often combined with a scaffold or biomaterial, is used in tissue engineering applications. Researchers are also developing local delivery systems using hydrogels or nanoparticles that can release cells or their secretions in a controlled way at a specific site.
The challenge in all of these approaches is ensuring that enough cells survive the delivery process, that they migrate to the right location, and that they remain active for long enough to produce a therapeutic effect.
Challenges, Risks, and Limitations
The promise of stem cell therapy is real, but so are the obstacles. Understanding both is essential for anyone trying to make sense of what they read about this field.
Scientific Obstacles
Even when stem cell transplants are performed, many of the transplanted cells do not survive. Poor engraftment, which refers to the failure of transplanted cells to successfully establish themselves in the recipient’s body, is a persistent problem. Researchers are working to improve survival rates by modifying the cells before transplantation or by preparing the recipient tissue more effectively.
Controlling differentiation with precision is another challenge. Coaxing stem cells to become the specific cell type needed, in the right proportions, and without producing unwanted cell types alongside them, requires extraordinary precision. When pluripotent cells are not fully guided through the differentiation process, they can form teratomas, tumors that contain a chaotic mix of different tissue types. Preventing this is one of the primary safety considerations in any therapy that uses pluripotent cells.
Manufacturing is also a significant issue. Moving from laboratory-scale experiments to producing clinical-grade cells in the quantities needed to treat large numbers of patients requires sophisticated facilities and quality control processes that are expensive and technically demanding.
Regulatory and Safety Considerations
The regulatory path for stem cell therapies is genuinely complex. In the United States, the FDA oversees cell therapies as biological products and requires companies to demonstrate safety and effectiveness through a staged clinical trial process before a therapy can be approved for general use. The same principle applies in most other developed countries, though specific regulations vary.
The approval process is deliberately rigorous and slow. This is because stem cell therapies involve living cells that can behave unpredictably in the body, and the long-term consequences of some interventions are not yet fully understood. There is a real gap between what researchers have shown in early trials and what is actually approved for routine clinical use, and it is important for patients and the public to understand that gap.
The Problem of Unregulated Clinics
Perhaps the most urgent practical concern in the stem cell field right now is the proliferation of clinics offering unproven stem cell treatments for a remarkable variety of conditions, often for very large sums of money.
These clinics typically advertise online, claim their treatments can address everything from arthritis to autism to Parkinson’s disease, and operate outside of any clinical trial framework. They present no peer-reviewed evidence for their effectiveness, and in many cases their treatments have caused documented harm, including infections, vision loss, tumor growth, and death.
The business model preys on people who are desperate for relief from serious conditions and may not have the tools to evaluate medical claims critically. Regulatory agencies have taken action against some of these clinics, but the problem has not gone away.
Any patient considering a stem cell treatment should ask several questions: Is this therapy approved by a recognized regulatory authority? Is it being offered as part of a registered clinical trial? Is there published peer-reviewed data on its safety and effectiveness? Anyone offering a stem cell therapy without satisfying these criteria deserves serious skepticism.
The Ethics of Stem Cell Research
The ethical questions in this field go beyond the embryo debate, though that remains a significant one.
The Embryo Question
The destruction of embryos to harvest embryonic stem cells is the issue that has generated the most public controversy. The core disagreement comes down to when in development a human organism acquires moral status deserving of protection.
Those who believe human life begins at fertilization argue that embryos, even at the blastocyst stage before implantation, should not be destroyed for research purposes regardless of potential medical benefits. Those who take a different view point out that the embryos used in research are typically surplus embryos from fertility clinics that will be discarded anyway, and that allowing them to contribute to research that could alleviate enormous suffering is actually the more ethical course.
This debate has not been resolved, and it is unlikely to be resolved soon, because it reflects genuinely different underlying values rather than a factual disagreement that new evidence could settle. The development of iPSC technology has shifted some attention away from embryonic stem cells, but it has not ended the debate, partly because ESC research continues for good scientific reasons and partly because iPSC research itself raises its own set of ethical questions.
Cloning and Its Implications
A technique called somatic cell nuclear transfer, or SCNT, involves taking the nucleus from an adult cell and inserting it into an egg cell from which the nucleus has been removed. The result can develop into an embryo genetically identical to the adult cell donor. When performed with the goal of producing an embryo from which stem cells can be extracted, this is called therapeutic cloning.
The distinction between therapeutic cloning and reproductive cloning, which would involve implanting the embryo and allowing it to develop into a person, is scientifically clear but ethically fraught. Critics worry that permitting therapeutic cloning creates the knowledge and infrastructure that could facilitate reproductive cloning, and most countries have prohibited reproductive cloning even if they permit some forms of therapeutic research.
Access, Equity, and Commercialization
Stem cell therapies that are already approved, like certain bone marrow transplants, are expensive and not equally accessible around the world. The emerging therapies in development are likely to be even more expensive when they reach the clinic. iPSC-based personalized therapies, which require manufacturing cells specific to each individual patient, could be extraordinarily costly.
This creates real questions about who benefits from the science. If regenerative medicine produces breakthroughs that are only available to those who can afford them, the field’s overall impact on human health will be far smaller than its advocates hope.
Cord blood banking raises a related equity issue. Private cord blood banking is marketed heavily to expectant parents but costs hundreds of dollars upfront plus ongoing storage fees, putting it out of reach for many families. Public cord blood banks, which collect and store donations for anyone who might need them, are a less commercially promoted but arguably more equitable alternative.
There are also important questions about intellectual property and informed consent. Stem cell lines developed from donated cells have been patented and commercialized, sometimes without the donors fully understanding how their donated material would be used. Ensuring that people understand exactly what they are consenting to when they donate biological material for research is an ongoing priority in bioethics.
The Future of Stem Cells and Regenerative Medicine
Research Directions Worth Watching
The combination of stem cell biology with gene editing technology, particularly CRISPR-Cas9, is opening up possibilities that would have seemed like science fiction even a decade ago. Researchers can now edit the genome of stem cells with considerable precision before transplanting them, which could allow them to correct genetic defects, improve the cells’ survival after transplantation, or remove the markers that trigger immune rejection. Early clinical trials combining iPSC therapy with gene editing are already underway for certain blood disorders.
Universal donor cell lines are another area of active development. The goal is to create stem cell lines that have been engineered to evade immune recognition, making them usable in any patient without requiring immune suppression. Several biotech companies are pursuing this, and the potential to produce off-the-shelf cellular treatments could dramatically reduce the cost and complexity of stem cell therapy.
Three-dimensional bioprinting, which uses stem cells as a kind of biological ink to print tissue structures layer by layer, is advancing quickly. Researchers have printed functional patches of heart tissue, liver tissue, and even simple vascular structures. Printing a complete organ remains far beyond current capability, but the field is moving faster than most expected.
Cell-free approaches, which deliver extracellular vesicles and other stem cell secretions rather than the cells themselves, avoid many of the safety and manufacturing challenges of cell-based therapy. These are being investigated for wound healing, neurological conditions, and immune regulation, among other applications.
From Lab to Clinic: The Clinical Trial Process
It is worth being clear about how a stem cell therapy actually moves from a research discovery to an approved treatment, because the timeline is long and the process is demanding.
After preclinical work in cells and animals suggests a therapy might be safe and effective, human clinical trials proceed in phases. Phase I trials focus primarily on safety and typically involve a small number of patients with few or no good existing treatment options. Phase II trials test effectiveness more rigorously in a larger group. Phase III trials involve large numbers of patients compared against existing treatments or placebo and are designed to produce the statistical confidence needed for regulatory approval. This entire process can take 10 to 20 years and cost enormous amounts of money, and most candidates fail somewhere along the way.
Anyone interested in participating in a clinical trial can search for registered trials through ClinicalTrials.gov in the United States or similar registries in other countries. Participation in legitimate trials is one of the most meaningful ways patients can contribute to advancing the field.
A Realistic Timeline
In the near term, meaning the next one to five years, the most likely advances will be expansions of already-approved approaches: new applications for HSC transplants, further refinements in limbal stem cell therapy, and regulatory approvals for a small number of MSC-based treatments that are already in late-phase trials.
Over the next five to fifteen years, the most advanced research programs targeting cardiac repair, Parkinson’s disease, diabetes, and macular degeneration may produce clinical approvals, assuming the ongoing trials continue to generate favorable data.
The more ambitious goals, lab-grown organs for transplant, routine personalized iPSC therapy, and comprehensive treatment of complex neurological conditions, remain longer-term prospects. Fifteen to twenty-five years is a reasonable estimate, and even that depends on scientific progress that has not yet happened.
Putting It All Together
Stem cells are not a single thing or a single technology. They are a broad category of biological material with a remarkable range of properties, and the therapies being built around them reflect that diversity. Some applications, like bone marrow transplantation, have been standard medicine for generations. Others are early-stage experiments that may or may not pan out. Still others are well-intentioned but unproven treatments being offered by clinics that are not operating within the legitimate medical system.
Sorting through all of this requires a level of scientific literacy that the average person is not always taught how to apply. When a news article says “stem cells may hold the key to treating Alzheimer’s,” that is usually describing preliminary research, not an approved therapy. When a clinic’s website claims to treat twenty different conditions with stem cell injections, that should raise immediate red flags.
The legitimate science in this field is genuinely exciting. Researchers have been able to grow functional heart muscle from pluripotent stem cells. They have restored corneal vision. They have helped patients with blood cancers go into lasting remission. They have reprogrammed adult cells back to an embryonic-like state in a way that bypasses the need for embryos. These are real achievements.
But they are also the product of painstaking, expensive, carefully controlled work done by researchers who are honest about what they know and what they do not know. That carefulness and honesty is exactly what separates legitimate stem cell medicine from the clinics that are willing to take your money and offer you hope they cannot back up.
If there is one takeaway from this entire subject, it is that the gap between what stem cells can theoretically do and what they can safely and reliably do in actual patients is still very real. That gap is closing, year by year, trial by trial. Understanding that gap clearly is the first step to understanding this field, and to making good decisions about it.
Key Terms Glossary
Differentiation: The process by which a less specialized cell becomes a more specialized cell type with a specific function.
Engraftment: The successful establishment of transplanted cells in the recipient’s body, where they survive and begin functioning.
Extracellular vesicles: Tiny membrane-enclosed particles released by cells that carry proteins, lipids, and genetic material to other cells as signals.
Graft-versus-host disease (GvHD): A complication of allogeneic transplants in which donor immune cells attack the recipient’s tissues.
Hematopoietic stem cells (HSCs): Blood-forming stem cells found in bone marrow that produce all types of blood cells.
Induced pluripotent stem cells (iPSCs): Adult cells that have been reprogrammed in the laboratory to behave like pluripotent stem cells.
Mesenchymal stem cells (MSCs): Multipotent stem cells found in bone marrow and other tissues that can form bone, cartilage, and fat cells.
Organoids: Small, three-dimensional tissue structures grown from stem cells in the laboratory that mimic aspects of organ structure and function.
Paracrine signaling: A form of cell communication in which a cell releases molecules that affect neighboring cells.
Pluripotent: Capable of differentiating into any cell type in the body (but not placental tissue).
Potency: A measure of how many different cell types a stem cell can develop into.
Self-renewal: The ability of a stem cell to divide and produce daughter cells that are also stem cells.
Stem cell niche: The local environment surrounding a stem cell, including neighboring cells and signaling molecules, that regulates its behavior.
Teratoma: A type of tumor that can form from pluripotent stem cells and contains a disorganized mix of different tissue types.
Totipotent: Capable of developing into any cell type, including placental cells; describes the fertilized egg and cells from the earliest cell divisions.