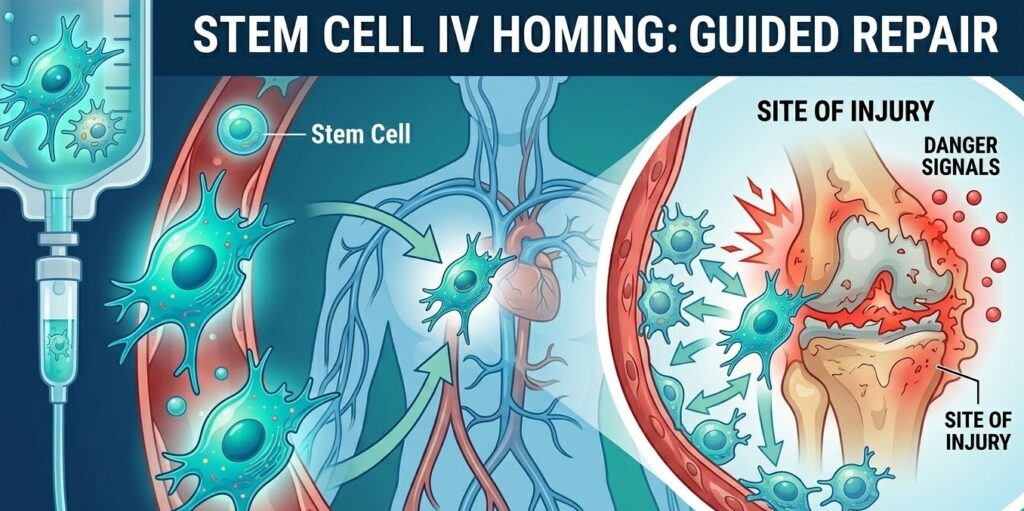
How IV Stem Cells Find the Site of Injury
When a doctor administers stem cells through an IV line, those cells enter the bloodstream at the arm and then — somehow — a meaningful number of them end up at a damaged knee, an inflamed joint, or an injured area of the brain. If you pause to think about that for a second, it really is remarkable. The human body contains roughly 60,000 miles of blood vessels. How does a cell injected at one point in that enormous network find its way to a very specific site of damage somewhere else entirely?
The answer is not luck. It is not random drift. What happens is a carefully coordinated biological process called stem cell homing, and understanding it helps explain why intravenous (IV) delivery of stem cells has become one of the most widely researched routes of administration in regenerative medicine. This process involves chemical signals, surface receptors, and a step-by-step sequence of molecular events that guides stem cells toward injured tissue with surprising precision.
This article breaks down that entire process in plain language, from the signals an injured tissue broadcasts to the way a stem cell physically crosses a blood vessel wall to reach its destination.
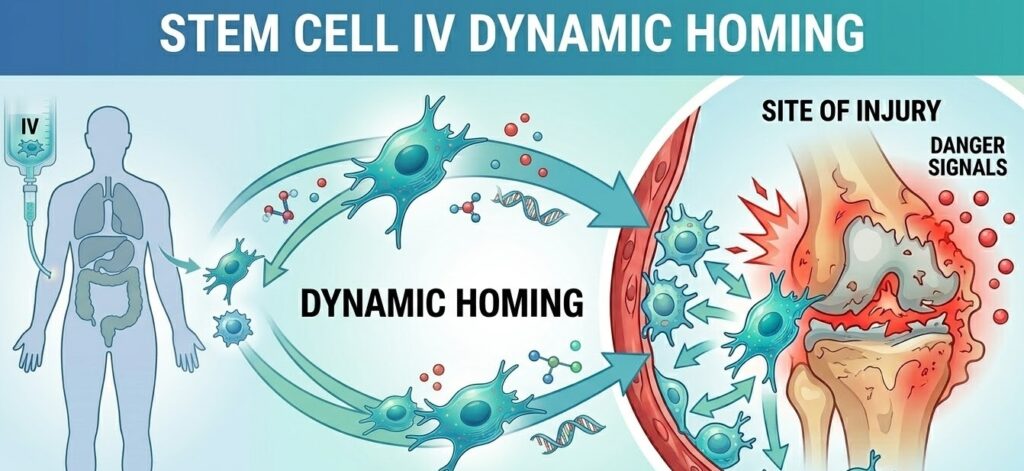
What Stem Cell Homing Actually Means
Homing, in the biological sense, refers to the ability of circulating stem cells to detect injury or inflammation and migrate toward it. The term was borrowed from the concept of a homing pigeon, which finds its way back to a fixed point no matter where it starts. In stem cell biology, the “fixed point” is not predetermined but is instead defined in real time by whatever tissue is currently damaged or under stress.
Stem cell homing has been studied extensively since researchers first noticed that transplanted bone marrow cells could travel through the bloodstream and settle in organs experiencing injury. Early work focused on hematopoietic stem cells, the type responsible for replenishing blood and immune cells. Researchers eventually discovered that mesenchymal stem cells (MSCs), which are derived from bone marrow, adipose (fat) tissue, and umbilical cord tissue, also possess homing capabilities, though the mechanisms differ slightly.
It is important to draw a distinction between active homing and passive accumulation. Passive accumulation happens when cells get mechanically trapped in narrow capillaries simply because of their size. Active homing is something more sophisticated. It involves the stem cell responding to biochemical cues, changing its surface chemistry, and navigating through tissue with directionality. Both can occur after an IV infusion, but it is the active homing mechanism that holds the most therapeutic promise.
At clinics like Stem Cells Colombia, the practical application of these biological principles informs how IV treatments are designed and timed, particularly in terms of ensuring that enough cells are in a state ready to respond to injury signals when they enter circulation.
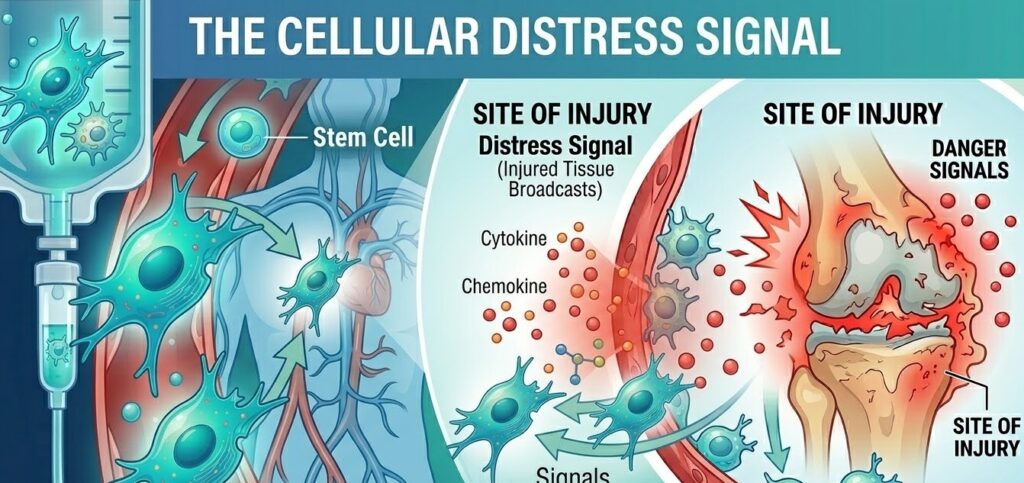
The Distress Signal: What Injured Tissue Broadcasts
Before a stem cell can home anywhere, the target tissue has to announce that something is wrong. Injured or inflamed tissue does this by releasing a collection of small signaling proteins called cytokines and chemokines. These molecules diffuse outward from the injury site and eventually reach the blood vessel walls nearest to the damage, creating a concentration gradient that cells in circulation can detect.
One of the most important of these molecules is a chemokine called SDF-1, also known as CXCL12. SDF-1 is produced in large quantities by damaged tissue, activated immune cells, and the endothelial cells lining blood vessels near the injury. Its concentration is highest right at the injury site and decreases the farther you move away, creating a gradient that acts almost like a trail of breadcrumbs.
Other signaling molecules contribute to the distress signal as well. Tumor necrosis factor-alpha (TNF-alpha) and interleukins such as IL-1beta, IL-6, and IL-8 are all elevated following tissue injury and promote what researchers call a pro-inflammatory microenvironment. This environment is actually necessary for homing to occur efficiently. It may seem counterintuitive that inflammation helps, but the inflammatory state is precisely what makes the blood vessel walls near the injury “sticky” and permeable enough for stem cells to adhere and cross through.
Injured tissue also releases molecules known as damage-associated molecular patterns, or DAMPs. These are molecular fragments released when cells rupture or die, and they act as danger signals that alert both the immune system and circulating stem cells that a repair response is needed. Additionally, areas of tissue damage typically have lower oxygen levels than healthy tissue, a condition called hypoxia. Hypoxic tissue upregulates a protein called HIF-1alpha (hypoxia-inducible factor 1-alpha), which in turn drives the production of even more SDF-1. So the hypoxic state and the chemokine signal reinforce each other, making the distress beacon stronger the more severe the injury is.
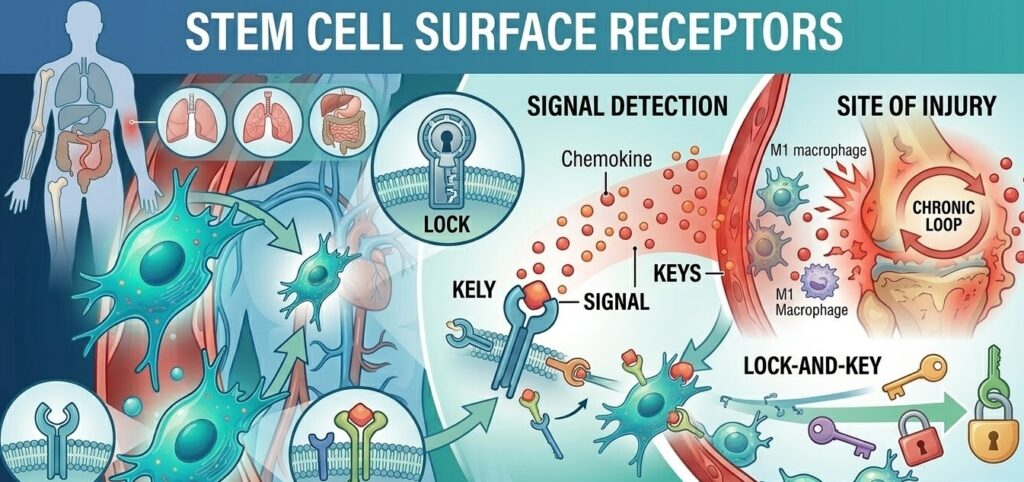
The Stem Cell’s Receiver: Surface Receptors
A stem cell cannot respond to a chemical signal unless it has a receptor designed to detect that specific signal. Think of it as a lock-and-key relationship. The injured tissue sends out the key (the chemokine), and the stem cell needs the right lock (the receptor) to respond.
The most critical receptor for IV stem cell homing is CXCR4, which is the primary receptor for SDF-1/CXCL12. When SDF-1 binds to CXCR4 on the surface of a stem cell, it triggers a cascade of internal changes that activate the cell’s migration machinery. This CXCR4/SDF-1 axis is so fundamental to stem cell homing that researchers frequently refer to it as the body’s stem cell GPS system.
Other receptors matter too. CD44 allows stem cells to bind to hyaluronic acid, a component of the extracellular matrix that accumulates at injury sites. Integrins, particularly VLA-4 and LFA-1, are surface proteins that help stem cells grab onto the inner wall of blood vessels and begin the process of crossing into tissue. Integrin activation is a key step in the transition from a freely circulating cell to one that has made firm contact with the vessel wall.
One important clinical consideration is that CXCR4 expression is not always high on stem cells by the time they are prepared for infusion. Cells that have been cultured for many passages in a laboratory setting can lose some of their surface receptor expression, which means their homing ability may be reduced. This is one reason why the preparation process matters, and why researchers have explored pre-conditioning strategies to boost receptor levels before administration.
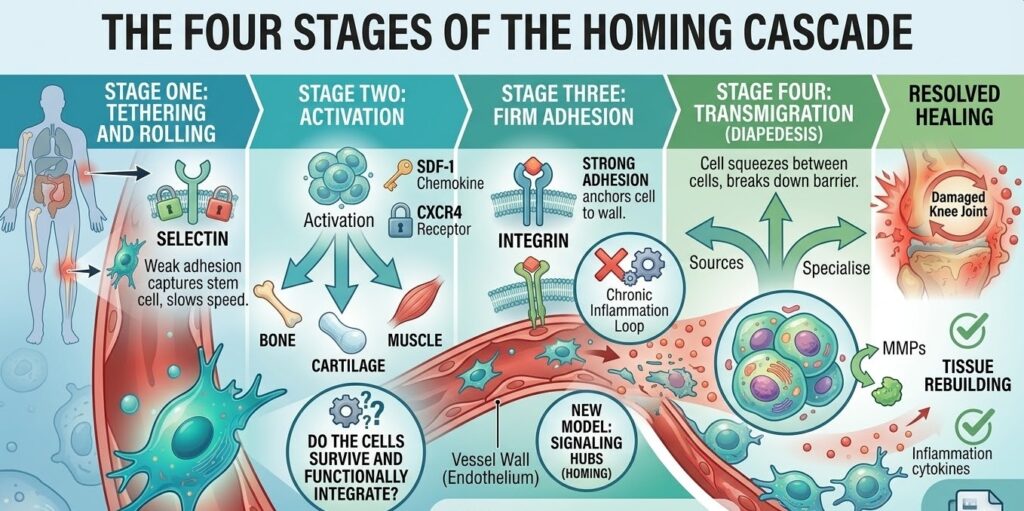
The Four Stages of the Homing Cascade
The process by which a stem cell moves from free circulation in the bloodstream to the interior of an injured tissue follows a specific sequence. Researchers have broken this down into four stages, each dependent on the one before it.
Stage One: Tethering and Rolling
When a stem cell flows through a blood vessel near an injured area, it encounters blood vessel walls that have been activated by the local inflammatory signals. These activated endothelial cells begin expressing surface proteins called selectins, specifically P-selectin and E-selectin. Selectins act as weak adhesion molecules that briefly catch stem cells as they pass by. The cell does not stop immediately but instead slows down and begins rolling along the vessel wall, tethered loosely by repeated selectin-receptor interactions. This rolling dramatically reduces the cell’s speed and gives it time to sample the local chemical environment.
Stage Two: Activation
While rolling, the stem cell comes into contact with chemokines like SDF-1 that are displayed on the surface of the endothelial cells. These chemokines bind to CXCR4 and other receptors on the stem cell’s surface. This binding triggers a process called inside-out signaling, where the signal travels from the outside of the cell to the inside and causes a conformational change in the cell’s integrin proteins. Essentially, the integrins “switch on” and become capable of making much stronger bonds with the vessel wall. The cell is no longer just passively rolling. It is now actively preparing to stop.
Stage Three: Firm Adhesion
Once the integrins are activated, they bind tightly to proteins on the endothelial surface, particularly VCAM-1 and ICAM-1, which are upregulated during inflammation. This firm adhesion arrests the rolling cell completely. It is now anchored to the vessel wall, positioned at the point closest to the injury signal.
Stage Four: Transmigration (Diapedesis)
The final stage is the most dramatic. The stem cell physically squeezes through the junctions between endothelial cells to exit the bloodstream and enter the surrounding tissue. This process is called transmigration or diapedesis. To complete it, the cell secretes enzymes called matrix metalloproteinases (MMPs) that break down the basement membrane, a thin layer of connective tissue just beneath the blood vessel wall. Once through the basement membrane, the stem cell is in the tissue proper and can begin migrating toward the peak concentration of the injury signal.
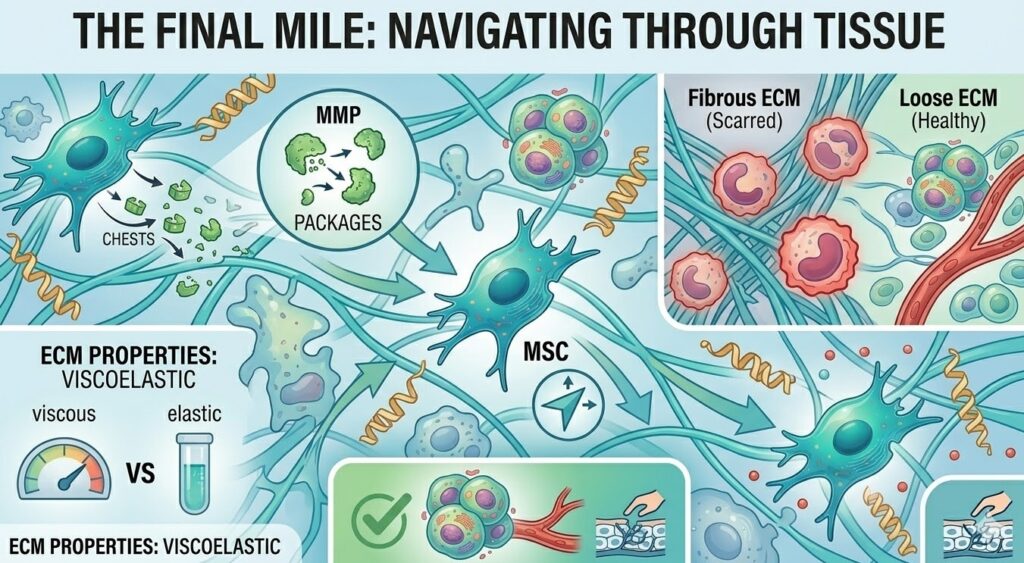
The Final Mile: Navigating Through Tissue
Exiting the bloodstream is not the end of the journey. The stem cell still has to travel through the extracellular matrix (ECM), the dense network of proteins and sugars that fills the spaces between cells. This is not easy going. The ECM is thick, viscoelastic, and variable in composition depending on the tissue type and how much damage has occurred.
The SDF-1/CXCR4 guidance system continues to operate during this phase. The stem cell follows the increasing concentration of SDF-1 as it moves deeper toward the injury core, a process called chemotaxis. It extends protrusions called lamellipodia in the direction of higher chemokine concentration and essentially pulls itself forward through the matrix.
One factor that can impede this final migration is fibrosis, or scar tissue. Chronic injuries that have been present for a long time often have dense, disorganized ECM with altered chemokine profiles. This can reduce the directional clarity of the signal and slow down the cell’s progress. This is one of the reasons that acute injuries tend to produce better homing outcomes than very old, chronic ones.
Why Not All Cells Reach the Target
A straightforward and honest question to address here is: do all the stem cells in an IV infusion actually reach the injured tissue? The answer is no, and it is worth understanding why.
A significant portion of intravenously administered stem cells are captured in the lungs before they ever reach the rest of the body. This is called the pulmonary first-pass effect, and it occurs because the lungs contain dense capillary beds with very narrow diameters. MSCs in particular are relatively large cells, and many of them get physically lodged in pulmonary capillaries. Studies have shown that a substantial fraction of IV-administered MSCs are retained in the lungs within the first few minutes after infusion.
This is not necessarily a total waste. Stem cells that are trapped in the lung can still have systemic effects through paracrine signaling, meaning they release bioactive molecules that enter circulation and exert anti-inflammatory and regulatory effects throughout the body. However, for conditions where you specifically want cells to reach a joint, the brain, or another distant organ, the pulmonary first-pass effect represents a real limitation.
Beyond the lungs, additional cells are filtered out by the liver and spleen, which are the body’s natural filtration organs. Researchers estimate that only a small percentage of IV-administered stem cells successfully home to the target tissue in most studies. This makes optimizing every step of the homing cascade critically important.
Factors That Affect How Well Cells Home
Not all stem cell infusions produce the same degree of homing, and several factors determine how efficiently the process works for a given patient and treatment.
Patient health and age play a significant role. Older patients and those with chronic metabolic conditions like diabetes or obesity often have altered cytokine environments that can impair the homing signal. The role of aging on stem cell function has been an active area of research, with findings suggesting that both the cells themselves and the tissue environment become less efficient at coordinating homing responses over time.
Cell source matters as well. Stem cells derived from umbilical cord tissue, adipose tissue, and bone marrow each have somewhat different surface receptor profiles, which translates to differences in homing efficiency for different tissue types. Umbilical cord-derived MSCs tend to have higher CXCR4 expression than adipose-derived cells, which may give them an advantage in certain applications.
The timing of treatment relative to the injury is another major variable. The inflammatory window, meaning the period when chemokine signals are at their strongest, typically occurs in the subacute phase of an injury, days to a few weeks after the initial event. Administering cells during peak inflammation gives them the strongest signal to follow. Treating very old, chronic conditions may still produce benefits, but the homing signal is often weaker and less directional.
Cell preparation quality is a factor that patients may not think to ask about but is critically important. Cells that have been cultured through many laboratory passages can lose receptor expression and viability. The way cells are processed, stored, and prepared for infusion can meaningfully affect how many of them arrive with functional CXCR4 and integrin profiles.
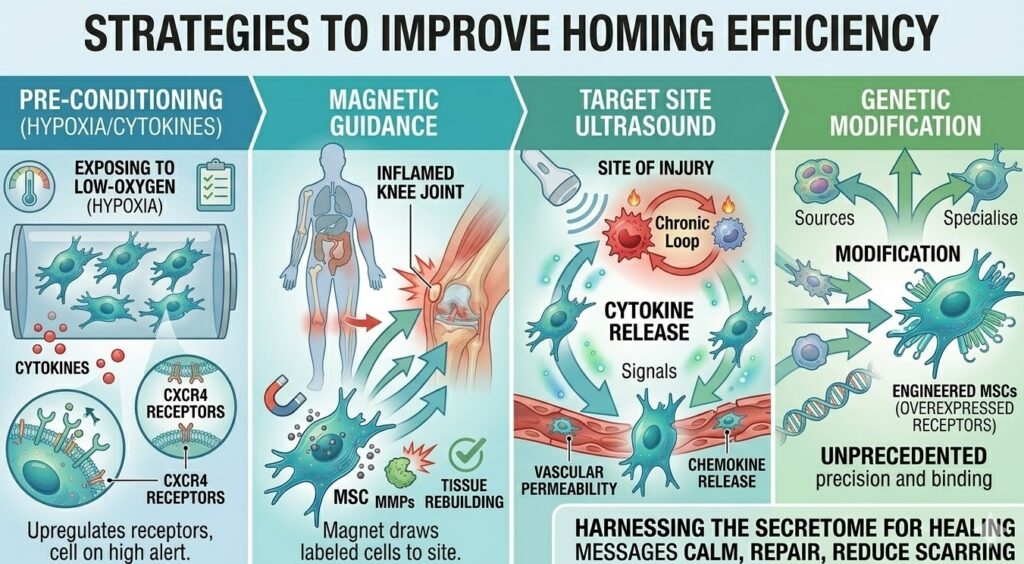
Strategies to Improve Homing Efficiency
Researchers are not content to accept the natural inefficiency of IV stem cell delivery. A range of strategies are being investigated to improve how reliably cells reach their targets.
Pre-conditioning is one of the most straightforward approaches. Exposing stem cells to a low-oxygen environment (hypoxia) before infusion has been shown to upregulate CXCR4 expression, essentially putting the cells on high alert before they enter the body. Treating cells with certain cytokines or pharmacological agents before infusion can have a similar effect.
Magnetic guidance is a more experimental approach in which stem cells are loaded with iron oxide nanoparticles and an external magnet is applied over the target tissue after infusion. The magnetic field draws labeled cells toward the surface of the body closest to the magnet, effectively concentrating them in the vicinity of the target. This approach has been tested in preclinical models with promising results but faces challenges in deep-tissue applications.
Ultrasound applied to the target site can temporarily increase vascular permeability and local chemokine release, making it easier for circulating cells to transmigrate at that location. This concept of creating an “artificial homing depot” is still being refined but represents an interesting approach to solving the precision problem.
Genetic modification of stem cells to overexpress homing receptors has also been explored in research settings. Engineering MSCs to have higher CXCR4 expression consistently improves their homing efficiency in animal models. The translation of this approach to clinical use involves additional regulatory and safety considerations.
What Happens After Homing
Homing is the beginning of the therapeutic process, not the end. Once a stem cell reaches the injured tissue, what happens next depends heavily on the local environment it finds there.
Most transplanted stem cells do not permanently engraft into the tissue in large numbers. The current scientific understanding is that the primary mechanism of benefit is paracrine signaling, meaning the cells release a wide array of bioactive molecules including growth factors, anti-inflammatory cytokines, and extracellular vesicles that influence the behavior of the surrounding native cells. This paracrine model helps explain why relatively small numbers of engrafted cells can still produce meaningful therapeutic effects.
The cells that do home successfully also interact with the local immune environment, often shifting it from a pro-inflammatory state toward one that is more conducive to repair. This immunomodulatory effect is one of the most well-supported benefits of MSC therapy and is closely tied to the homing process, since cells that never reach the injury cannot directly modulate the local immune response.
What This Means If You Are Considering IV Stem Cell Therapy
Understanding the homing mechanism gives patients a more grounded perspective on what IV stem cell therapy can and cannot do. The cells are not simply injected and then magically distributed everywhere. They follow a specific biological process, and that process has real variables that affect the outcome.
It is worth asking your provider questions like: What is the source of the cells? How many passages have the cells been through? What is the CXCR4 expression profile of the product? Is there any pre-conditioning protocol to enhance homing? These questions are reasonable and any serious clinic should be able to address them.
It is also worth having realistic expectations about numbers. Not every infused cell reaches the target tissue. The paracrine effects from cells that do not directly home to the injury still contribute to the overall response, but the cells that successfully transmigrate and reach the injury site are the ones most directly involved in local repair. Timing the treatment to coincide with an active inflammatory window, when chemokine signals are strongest, is one of the most practical ways to improve outcomes.
The Science Is Still Moving Forward
Research on stem cell homing is one of the most active areas in regenerative medicine. The past two decades have produced a much clearer picture of the molecular machinery involved, and that understanding is being applied to develop better cell products, smarter delivery strategies, and improved patient selection criteria.
What is clear from this body of research is that IV delivery of stem cells is not guesswork. The cells do not wander randomly until they stumble upon an injury. They follow a coordinated, multi-step cascade driven by real biochemical signals and precise receptor interactions. The homing mechanism is genuinely sophisticated, and the fact that it works at all, given the complexity of the task, is a testament to the elegance of human biology.
As researchers continue to optimize each step of the process, from enhancing the distress signals released by injured tissue, to improving receptor expression on administered cells, to reducing pulmonary trapping, the gap between IV delivery and direct injection is narrowing. For patients and providers, that progress translates into increasingly reliable and targeted therapeutic outcomes.