The Body’s Built-In Repair Problem
When you cut your finger, your body springs into action almost immediately. Within seconds, blood rushes to the area, swelling starts, and the familiar redness and warmth of inflammation begins. For a long time, scientists and doctors viewed inflammation as simply the first step in a predictable healing process — something that shows up, does its job, and then fades away as new tissue forms.
But that is not always how it works.
For millions of people, inflammation does not fade. It lingers, spreads, and ends up doing more harm than good. Chronic inflammatory conditions like rheumatoid arthritis, Crohn’s disease, multiple sclerosis, and some forms of heart disease are, at their core, cases where the body’s own immune response gets stuck in a loop it cannot escape. The fire meant to protect the body keeps burning after there is nothing left to fight.
This is where stem cell research has started to change how scientists think about healing. For years, the big promise of stem cells was their ability to turn into other types of cells — to grow into new heart muscle, fresh cartilage, or functioning neurons. That ability is real, but it turns out to be only a small part of what stem cells actually do in the body. The more scientists study them, the more they realize that stem cells are less like spare parts and more like a sophisticated communication system — one that sends chemical signals telling the immune system to calm down, telling damaged tissue to start rebuilding, and telling the body, in effect, that it is time to heal.
Understanding how that communication works is one of the most compelling areas of biomedical research happening right now.
What Inflammation Actually Is and Why It Goes Wrong
Before getting into how stem cells affect inflammation, it helps to understand what inflammation is and what it is supposed to do.
When the body detects an injury or an infection, the immune system floods the affected area with specialized cells and chemical messengers. These messengers include proteins called cytokines — specifically pro-inflammatory cytokines like tumor necrosis factor alpha (TNF-alpha), interleukin-1 beta (IL-1beta), and interleukin-6 (IL-6). Their job is to recruit more immune cells to the area, raise the local temperature to slow bacterial growth, increase blood flow, and trigger the physical symptoms we associate with inflammation: redness, swelling, heat, and pain. You can read more about how this process works on the National Institute of Allergy and Infectious Diseases website.
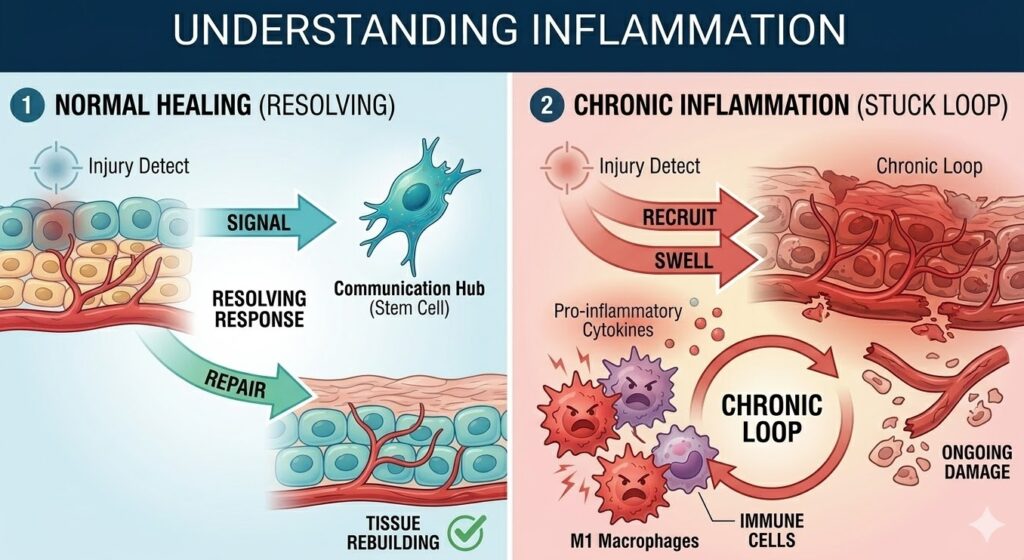
This response is not a malfunction. It is the body doing exactly what it evolved to do. The problem starts when this response does not resolve properly. In a healthy healing process, anti-inflammatory signals eventually take over, immune activity winds down, and tissue repair begins. Researchers refer to this winding-down phase as the resolution of inflammation, and it is a tightly regulated process with its own set of chemical signals. A detailed review of this resolution process was published in Nature Reviews Immunology, which outlines just how many molecular steps are involved in turning off an immune response properly.
When resolution fails, the body gets stuck in a state of chronic inflammation. Immune cells keep attacking, cytokines keep signaling for more immune activity, and the original tissue suffers ongoing damage. Over time, this leads to the kind of destruction seen in rheumatoid arthritis, where joints are slowly worn away, or in inflammatory bowel disease, where the lining of the digestive tract is repeatedly damaged and scarred. The Centers for Disease Control and Prevention estimates that chronic diseases linked to inflammation affect over 60% of adults in the United States.
The challenge with treating chronic inflammation is that most available drugs work by broadly suppressing the immune system. While that can reduce symptoms, it also leaves the body vulnerable to infections and does not fix the underlying failure of the resolution process. This is why researchers have been searching for more targeted approaches — and why stem cells have attracted so much attention.
What Stem Cells Are and Where They Come From
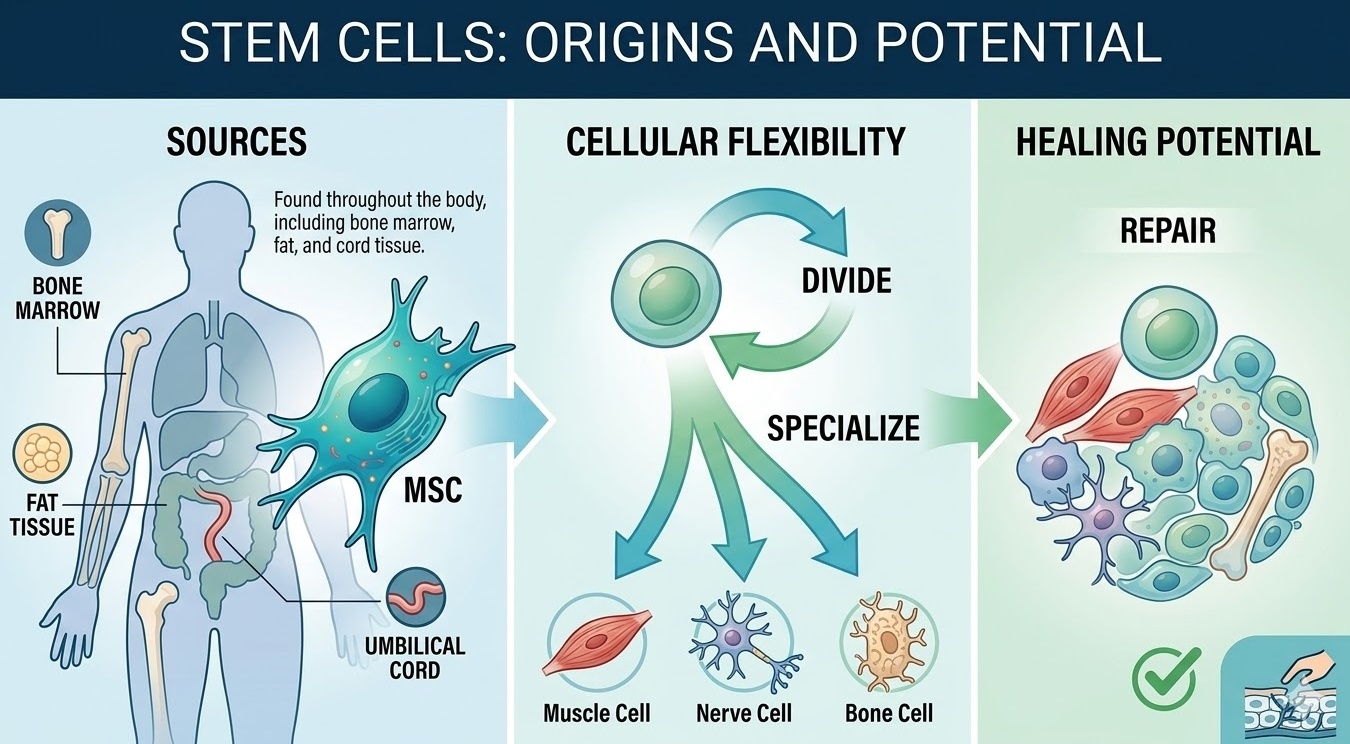
A stem cell is, at its most basic, a cell that has not fully committed to becoming a specific type of cell. It retains the ability to either continue dividing or to differentiate into more specialized cell types. This flexibility is what makes stem cells so important to both development and repair.
There are several types of stem cells, but the ones most relevant to inflammation and healing are mesenchymal stem cells, often abbreviated as MSCs. Mesenchymal stem cells are found throughout the body, with especially high concentrations in bone marrow, adipose (fat) tissue, the umbilical cord, and dental pulp. They were originally identified as cells that could become bone, cartilage, and fat, but research over the past two decades has revealed that their most important role in healing may have nothing to do with differentiation at all. A thorough overview of MSC biology is available through the National Center for Biotechnology Information.
Hematopoietic stem cells (HSCs), which live primarily in bone marrow and give rise to all blood and immune cells, are another important category. These are the cells used in bone marrow transplants for conditions like leukemia and lymphoma. Be The Match, the national bone marrow registry, provides accessible information on how these transplants work and who they may help.
Induced pluripotent stem cells (iPSCs), which are ordinary adult cells that have been reprogrammed back into a stem-like state, represent a third category that is rapidly gaining ground. The International Society for Stem Cell Research publishes guidelines on iPSC research and clinical translation that are considered the standard for responsible development in this area.
For most of the healing and anti-inflammatory effects discussed in this article, mesenchymal stem cells are the primary focus. They are the most extensively studied, and the research behind them is the most developed.
The Old Model Was Mostly Wrong
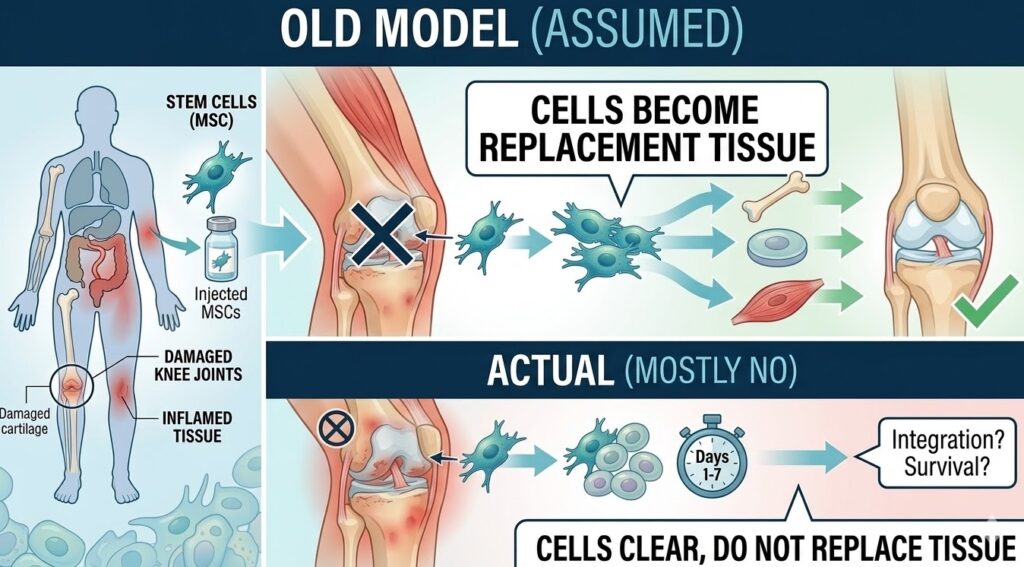
For a long time, scientists assumed that when stem cells were introduced into an injured area, they worked by traveling to the site, settling in, and literally transforming into the type of cell that had been damaged. In theory, you inject MSCs into a damaged knee, they become cartilage cells, and the knee heals. Simple enough.
The problem is that when researchers actually tracked what happened to transplanted stem cells, they found that the vast majority did not survive for more than a few days or weeks. They did not successfully integrate into the tissue, and they did not transform into large numbers of replacement cells. Yet patients and animal models treated with these cells often showed significant improvements in healing and inflammation.
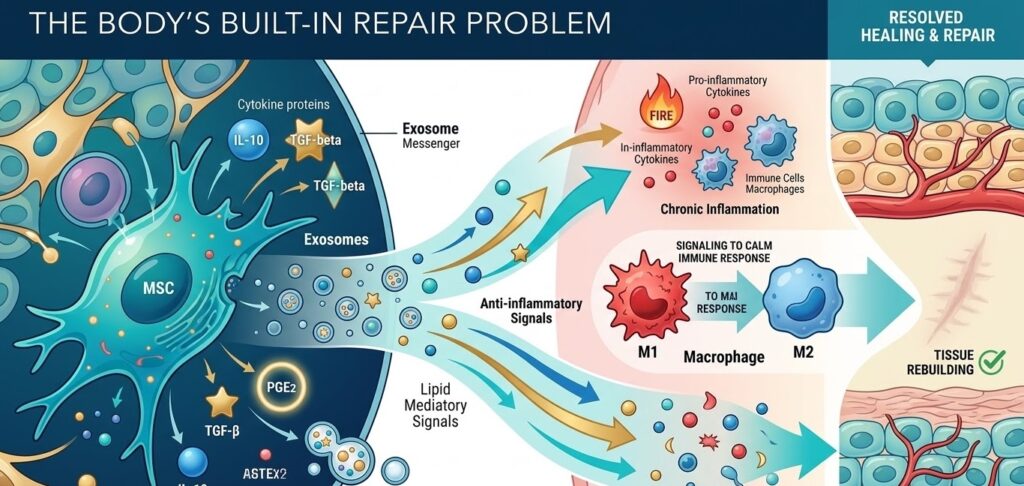
This disconnect forced a major rethinking of how stem cells work. The answer that emerged is called paracrine signaling — a mechanism where cells influence their neighbors not by becoming them, but by secreting bioactive molecules that change how those neighboring cells behave. Essentially, stem cells talk to the tissue around them, and that conversation triggers the healing response.
This shift in understanding has been one of the more important developments in stem cell biology. It means the goal is not necessarily to get stem cells to survive and integrate long-term. The goal is to get them to say the right things to the right cells at the right time.
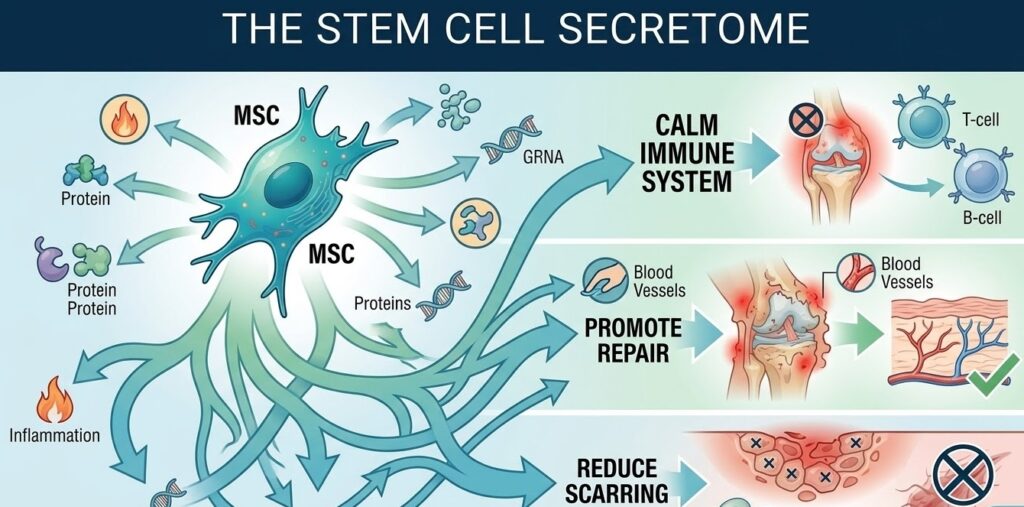
The Secretome: What Stem Cells Say to the Body
The collection of all the molecules a stem cell secretes is called its “secretome,” and it is remarkably complex. MSCs release a wide variety of proteins, lipids, and genetic material that collectively instruct the immune system, promote blood vessel growth, protect cells from dying, and reduce scarring. Research published in Frontiers in Immunology has catalogued many of the key components of the MSC secretome and their downstream effects on immune and tissue cells.
Some of the most important anti-inflammatory signals in the secretome include:
Interleukin-10 (IL-10): This is one of the body’s most powerful anti-inflammatory cytokines. When MSCs release IL-10, it suppresses the production of pro-inflammatory cytokines and dials down immune cell activity. It is a direct counter-signal to the TNF-alpha and IL-1beta that drive chronic inflammation.
Transforming Growth Factor-beta (TGF-beta): TGF-beta plays a dual role in healing — it dampens immune activation while also promoting tissue repair. It is particularly important for reducing the overactivity of T-cells, which are the immune cells responsible for tissue damage in many autoimmune diseases.
Prostaglandin E2 (PGE2): PGE2 is a lipid mediator that works on macrophages and T-cells, nudging them away from inflammatory behavior. It is the same compound that aspirin and ibuprofen inhibit, which is part of why those drugs reduce inflammation. MSCs produce it naturally as part of their response to inflammatory signals.
Hepatocyte Growth Factor (HGF): HGF promotes cell survival and has anti-fibrotic properties, meaning it helps prevent the formation of scar tissue that can permanently impair organ function after chronic inflammation.
Indoleamine 2,3-dioxygenase (IDO): IDO is an enzyme that breaks down tryptophan, an amino acid that certain immune cells need to proliferate. By depleting local tryptophan levels, MSCs can effectively put the brakes on runaway T-cell activity.
What makes this system impressive is that it is not static. Stem cells adjust what they secrete depending on the signals they receive from their environment. In a highly inflamed tissue, they respond to those inflammatory signals by ramping up their anti-inflammatory output. They are, in a very real sense, reading the situation and responding to it.
Exosomes: The Messenger Packets
One of the most exciting developments in this area is the discovery of the role that tiny particles called exosomes play in stem cell signaling. Exosomes are nano-sized vesicles — essentially tiny bubbles pinched off from the cell membrane — that carry proteins, lipids, messenger RNA, and microRNA from one cell to another. A comprehensive review of their biology and signaling roles was published in Nature Reviews Molecular Cell Biology and remains one of the most-cited papers in this area.
When MSCs release exosomes, those exosomes carry a concentrated payload of the same anti-inflammatory and pro-healing signals described above. They can travel through the bloodstream and reach distant tissues, and because of their small size and specific membrane composition, they can even cross the blood-brain barrier — the highly selective filter that normally keeps large molecules out of the brain.
This has opened up an entirely new avenue of research. Instead of transplanting live stem cells, researchers are studying whether purified MSC-derived exosomes could deliver the same therapeutic benefits. Dozens of clinical trials exploring exosome therapy are currently listed on ClinicalTrials.gov, covering applications from spinal cord injury to heart disease to kidney disease.
Beyond exosomes, stem cells also communicate through direct cell contact mechanisms like gap junctions and tunneling nanotubes. There is even emerging evidence that MSCs can physically transfer their own mitochondria — the energy-producing organelles inside cells — to damaged or stressed cells nearby, essentially lending their power supply to cells that are struggling to survive. This is a finding that is still being studied, but it suggests the communication system is even more sophisticated than researchers initially thought.
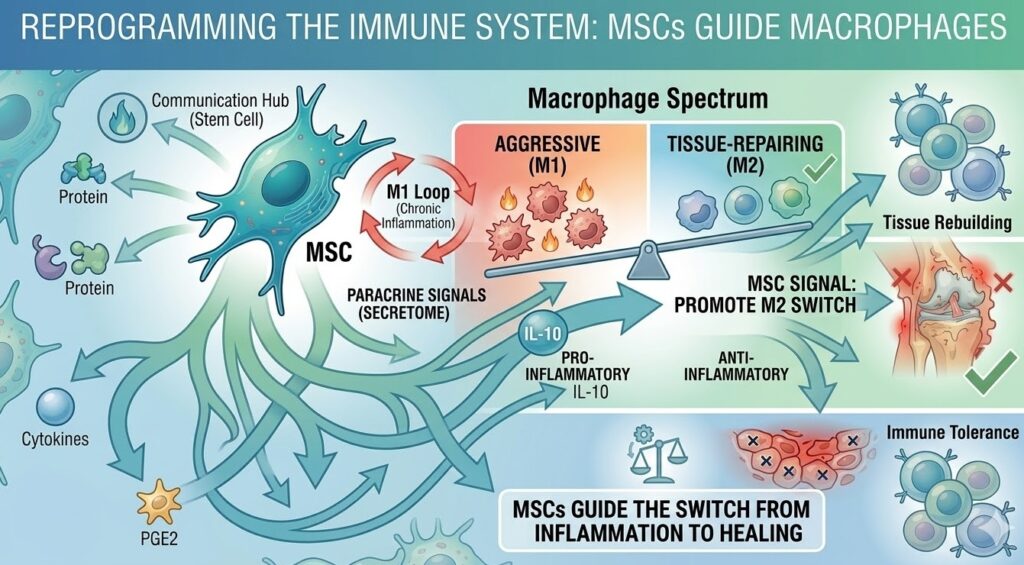
Reprogramming the Immune System
One of the most clinically significant things MSCs can do is change the behavior of macrophages. Macrophages are immune cells that come in two major functional types, often called M1 and M2, though the reality is more of a spectrum. M1 macrophages are aggressive and pro-inflammatory. M2 macrophages are anti-inflammatory and tissue-repairing. In a healthy healing process, there is a natural transition from M1 to M2 dominance as repair advances. In chronic inflammatory conditions, that transition stalls, and M1 macrophages stay in charge long past when they should have stood down. Research published through the NCBI database on macrophage polarization outlines the molecular details of this switch and how it can be influenced externally.
MSC signaling — particularly through PGE2 and IL-10 — actively promotes this M1-to-M2 transition. By changing the macrophage balance, stem cells help move tissue from a state of active inflammatory destruction toward one of active repair. This has been demonstrated in animal models of conditions ranging from cardiac injury to inflammatory bowel disease to traumatic brain injury.
MSCs also have significant effects on T-cells, which sit at the center of most autoimmune diseases. Specifically, they suppress the activity of Th1 and Th17 T-cell subsets while promoting the development of regulatory T-cells, often called Tregs. Tregs are sometimes described as the immune system’s peacekeepers — their job is to prevent excessive immune reactions and maintain tolerance to the body’s own tissues. The role of Tregs in autoimmune disease control is discussed in detail in research available through the National Institutes of Health.
Importantly, this immune modulation does not amount to broad immunosuppression. MSCs do not simply switch off the immune system the way corticosteroids or immunosuppressive drugs do. Instead, they promote a state of local immune tolerance — calming the overreaction without disabling the body’s ability to fight real threats. That distinction is clinically important, since broad immunosuppression carries serious risks of infection.
What Happens After the Inflammation Clears
Anti-inflammatory signaling is only part of the story. Once stem cells have helped calm the immune response, they also help set the conditions for actual tissue repair. Several molecules in the MSC secretome are directly pro-regenerative.
MSCs secrete VEGF (Vascular Endothelial Growth Factor), which stimulates the growth of new blood vessels — a process called angiogenesis. Restoring blood flow to damaged tissue is critical because oxygen and nutrients need to reach repair cells for tissue regeneration to move forward.
They also produce signals that suppress fibrosis, the process by which damaged tissue gets replaced by scar rather than functional cells. Fibrosis is a major problem in chronic inflammatory diseases — the scarring that results from long-term inflammation in the liver, lungs, kidneys, or heart can permanently compromise organ function. MSC-derived signals, particularly HGF and certain microRNAs carried in exosomes, interrupt the fibrotic signaling pathway and help preserve functional tissue.
In the brain and nervous system, MSCs secrete BDNF (Brain-Derived Neurotrophic Factor) and GDNF (Glial Cell Line-Derived Neurotrophic Factor), both of which support neuron survival and promote neural repair. These factors are being studied for applications in traumatic brain injury, spinal cord injury, Parkinson’s disease, and Alzheimer’s disease. A review of neurotrophin signaling and its therapeutic implications is available through the NCBI neurotrophin literature.
There is also a phenomenon called stem cell homing that deserves mention. When tissue is damaged, it releases chemical signals — including a molecule called SDF-1, which acts through a receptor called CXCR4 — that attract MSCs from bone marrow and other reservoirs toward the injury site. In a very real sense, the injured tissue is calling for help, and the stem cells are the ones answering.
Where This Is Being Applied Clinically
The research on MSC signaling has already translated into several clinical applications, though the field is still developing and the strength of the evidence varies significantly by condition.
Autoimmune diseases are among the most intensively studied applications. Hematopoietic stem cell transplants have been used to treat severe cases of multiple sclerosis and lupus with the goal of essentially resetting the immune system. The procedure involves eliminating the patient’s existing auto-reactive immune cells and rebuilding the immune system from transplanted stem cells. Research published by the NCBI covers the clinical outcomes of HSCT for autoimmune disease in considerable detail.
Graft-versus-host disease (GvHD) is one area where the evidence for MSC therapy is relatively strong. GvHD occurs when transplanted immune cells attack the recipient’s tissues after a bone marrow transplant. The FDA has approved an MSC-based product for treating steroid-refractory GvHD in pediatric patients, making it one of the more established regulatory approvals in this space.
Orthopedic applications are also widely used clinically. MSC injections for cartilage damage and osteoarthritis are being performed at many centers, though outcomes still show variability. The improvement seen in many patients is now understood to come primarily from anti-inflammatory signaling effects rather than from cartilage regeneration.
Neurological applications remain largely experimental, but the research is advancing. Studies in animal models of Parkinson’s, ALS, and spinal cord injury have shown meaningful effects, and a growing number of clinical trials exploring neural applications are registered on ClinicalTrials.gov.
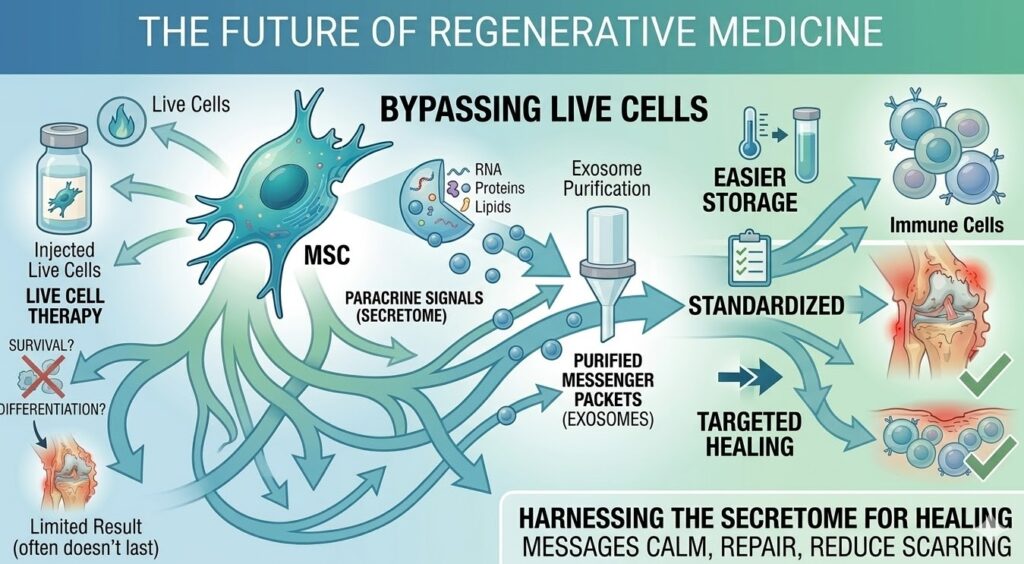
The Future of Stem Cell Signaling Therapy
The next wave of research is focused less on using stem cells themselves and more on harnessing what they produce. Exosome therapies — where the nano-particles secreted by stem cells are purified and delivered directly — are attracting significant interest because they sidestep many of the practical and safety challenges of live cell transplantation. Exosomes can be stored, standardized, and administered more easily than living cells.
Researchers are also working on ways to pre-condition stem cells before use — exposing them to low oxygen conditions, specific cytokines, or genetic modifications to amplify their anti-inflammatory output. The idea is to turn up the volume on the signals that matter most for a given condition.
Biomaterial scaffolds that can deliver stem cell-derived factors in a sustained, controlled way are another avenue being explored. Rather than injecting cells or exosomes that may disperse quickly, the idea is to place a scaffold loaded with therapeutic molecules directly at an injury site, allowing for prolonged local release.
Further ahead, synthetic biology approaches are being developed to engineer cells that produce specific anti-inflammatory signals only in response to specific triggers — essentially, programmable therapeutic cells that activate where and when inflammation reaches a certain threshold. Research in this area is being published regularly in journals like Nature Biotechnology and represents one of the longer-range frontiers in the field.
Proceed With Caution
None of this means that stem cell therapies are ready to replace conventional medicine, or that every clinic offering stem cell injections is operating on solid scientific ground.
There is a serious problem with unregulated clinics marketing unproven treatments for conditions ranging from autism to aging to sports injuries. The FDA has taken enforcement action against a number of these providers. Their consumer warning page on stem cell therapies is an important read for anyone considering a procedure that has not been part of a properly registered clinical trial.
There are also legitimate safety concerns. Introducing exogenous cells into the body carries some risk of unintended immune reactions. The variability in how MSCs behave depending on the donor, the tissue source, and the preparation method makes it difficult to standardize treatments and interpret research results across different studies.
What the field needs — and is actively working toward — is better standardization: agreed-upon methods for characterizing stem cell preparations, consistent dosing protocols, and more rigorous clinical trials with well-defined endpoints. None of these challenges make the science less promising. They just mean that moving from laboratory discovery to reliable clinical treatment takes time and care.
Conclusion
The idea that the body knows how to heal itself is not new. What is new is the growing understanding of just how central cell-to-cell communication is to that process — and how stem cells sit at the center of that communication network.
The old picture of stem cells as biological blank slates waiting to be stamped into replacement parts has given way to something far more interesting: a picture of highly responsive signaling hubs that read their environment, synthesize complex chemical messages, and transmit those messages to nearby and distant cells in ways that calm inflammation, protect tissue, rebuild the blood supply, and promote regeneration. They are not repair kits. They function more like diplomatic envoys working to restore peace after the immune system’s initial assault has done what it needed to do.
The clinical applications emerging from this research are real, even if the field still has significant growing to do. For patients with chronic inflammatory conditions, autoimmune diseases, and tissue injuries, this area of science offers genuine reason for hope — built on a growing body of evidence about how the body actually communicates with itself. The National Institute on Aging continues to fund research in this area, and the pace of discovery shows no sign of slowing.
If you are interested in how these breakthroughs in cellular signaling are being applied today, the medical team at Stem Cells Colombia is dedicated to bringing this research into a medically responsible clinical setting. Visit our homepage to learn more about our commitment to patient safety and scientific integrity.