If you have never heard of passage number before, you are not alone. It is one of those technical details that tends to stay inside the lab. But understanding it could change the questions you ask before any cell-based treatment, and it might change how you evaluate the quality of a clinic or provider.
What “Passage” Actually Means
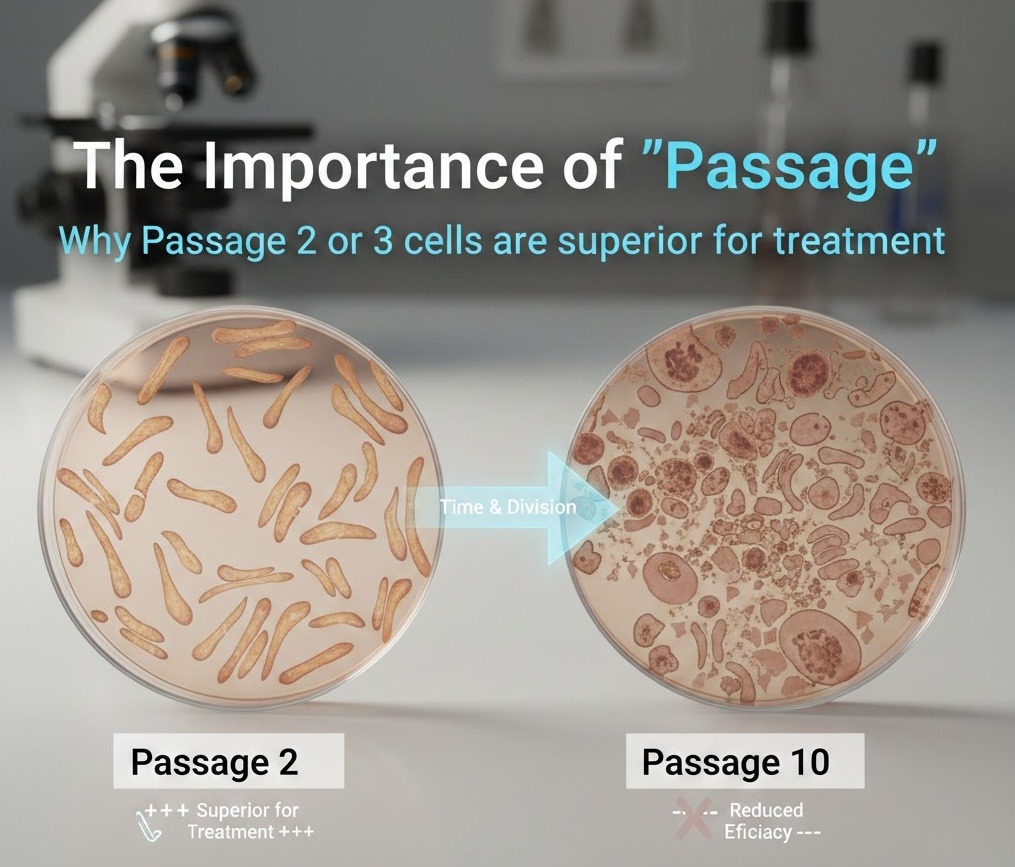
When cells are harvested from a donor tissue source, there are usually not enough of them to treat a patient right away. Scientists need to grow more cells in a laboratory setting, and they do this by placing the cells in a controlled environment where they can divide and multiply. Every time the cells outgrow their container and are transferred to a new one to keep expanding, that transfer is called a “passage.” The first time this happens, the cells become Passage 1, or P1. The second transfer makes them P2, then P3, and so on.
Think of it like making copies of a document. The first copy you make from the original looks almost identical. But if you then copy that copy, and copy that one, and keep going, you eventually start to notice small distortions accumulating. The text gets a little fuzzier, the alignment drifts slightly. The further you get from the original, the more quality you lose.
Cells work in a similar way. Every time they divide and are transferred, small changes accumulate. Most of those changes are minor in the early passages, but they compound over time. By the time you reach P6, P8, or higher, the cells you have in the lab may look and behave quite differently from the cells that were originally isolated.
This is why passage number matters so much. It tells you how many times cells have gone through this process, and therefore how close to or far from their original biological state they are.
Why Cells Are Expanded in the First Place
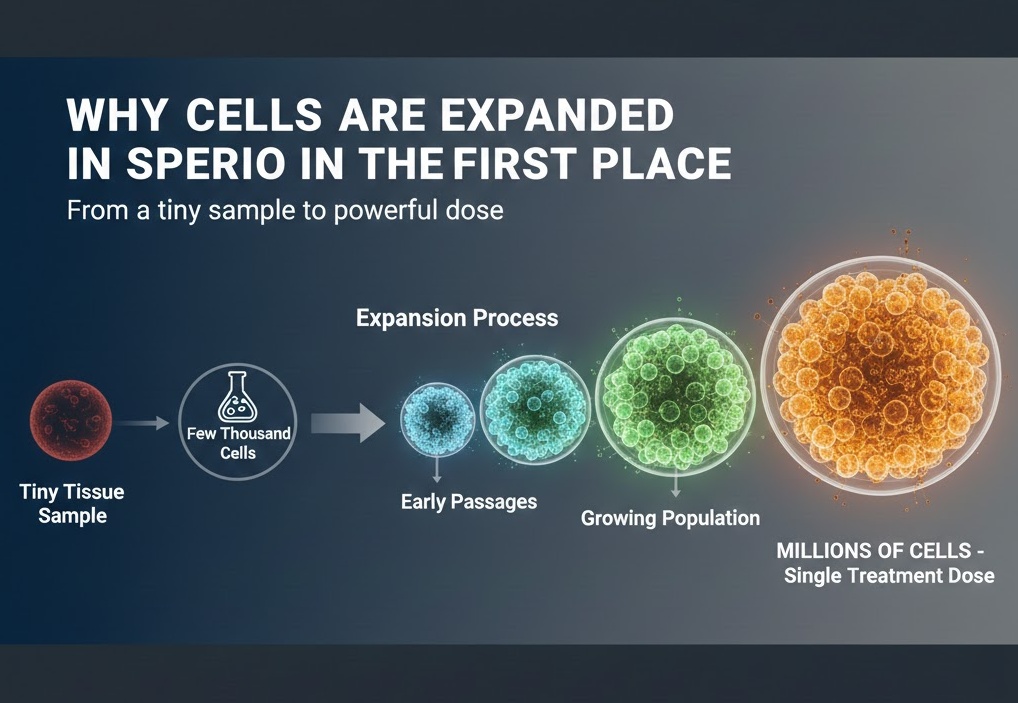
It is worth understanding why labs expand cells at all, because it is a legitimate and necessary part of producing any cell therapy. A tiny tissue sample might yield a few thousand to a few hundred thousand cells. A single treatment dose for most applications requires millions of cells. There is simply no way around the need to grow more.
This is true whether you are receiving a treatment in the United States, Europe, or accessing stem cell therapy in Colombia, where clinics often work with mesenchymal stem cells derived from umbilical cord tissue or adipose fat. No matter the source, the expansion process is unavoidable. The question is not whether cells are expanded, but how many times they are expanded before being used.
What Happens to Cells as Passage Number Climbs
The changes that occur across successive passages are not random or unpredictable. They follow patterns that researchers have studied in depth, and most of those patterns point in the same direction: higher passage means lower therapeutic quality.
Genetic Drift and Mutation Risk
Every time a cell divides, it has to copy its entire genome. This process is remarkably accurate, but it is not perfect. Over many division cycles, small errors called mutations can accumulate. In early passages, there have been so few rounds of replication that the risk of significant errors is low. But with each additional passage, the opportunity for those errors to stack up grows. Cells at high passage numbers carry a greater risk of having drifted from their original genetic blueprint, which affects everything from their behavior to their safety profile.
Telomere Shortening
Telomeres are protective caps on the ends of chromosomes, and they get a little shorter each time a cell divides. This is essentially a biological clock. When telomeres become too short, cells stop dividing effectively or begin to deteriorate. Cells that have been passaged many times have shorter telomeres, meaning they are closer to the end of their functional lifespan. When you receive high-passage cells in a therapeutic setting, you may be receiving cells that are already aging out of their most active and effective state.
Phenotypic Drift
“Phenotype” refers to the observable characteristics of a cell, including the proteins expressed on its surface and the signals it sends to surrounding tissue. These surface markers are how the body recognizes and responds to therapeutic cells. They are also how cells carry out their jobs, whether that means reducing inflammation, stimulating tissue repair, or modulating the immune system.
As cells are passaged, their phenotype can shift. Key surface markers may be expressed at lower levels or drop off entirely. The cells may start producing different cytokines, which are the signaling proteins they use to communicate with other cells in the body. In short, the cell’s functional identity can change, and not in ways that are therapeutically beneficial.
Senescence and the Bystander Effect
At some point, heavily passaged cells begin entering a state called senescence. Senescent cells are still alive, but they have essentially stopped dividing and started secreting a mix of inflammatory proteins. This is sometimes called the senescence-associated secretory phenotype, and it is a problem for therapy. Rather than helping reduce inflammation or support healing, senescent cells can actually promote inflammation in surrounding tissue. This is sometimes called the “bystander effect,” where cells that are no longer functional also negatively affect nearby healthy cells.
Epigenetic Changes
Beyond the DNA sequence itself, cells regulate which genes are active and which are silenced through a layer of chemical modifications called the epigenome. These modifications can change with repeated passaging. Genes that were active in early-passage cells, including genes associated with stem cell potency and tissue repair, may become silenced. Genes that were quiet may become active. These epigenetic shifts can fundamentally alter what the cells are capable of doing, even if the underlying DNA sequence looks the same.
Reduced Proliferative Capacity
When cells are administered to a patient, they need to be able to divide and establish themselves in the body, at least temporarily, to carry out their therapeutic effects. High-passage cells that are approaching senescence have a reduced ability to proliferate after administration. This limits their ability to migrate to sites of injury, integrate with local tissue, and perform the functions that make cell therapy worthwhile.

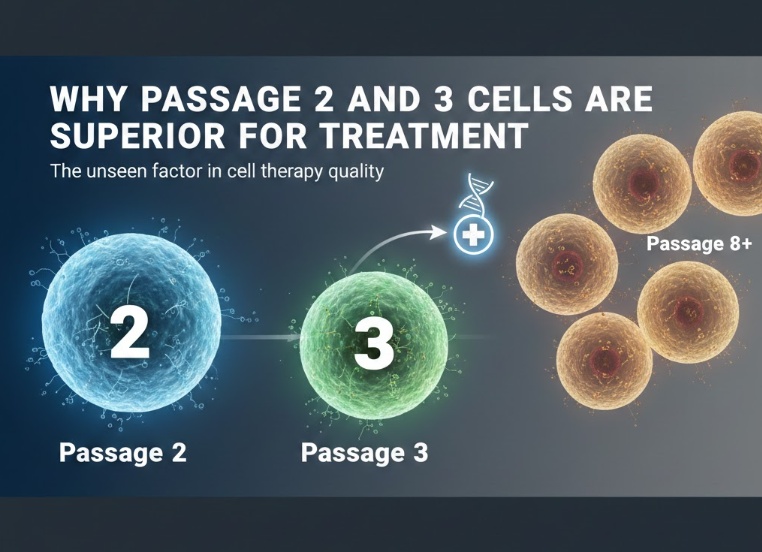
Why Passage 2 and 3 Are the Sweet Spot
Given everything that happens at higher passage numbers, it makes sense to use cells as early in the process as possible. So why not just use Passage 0 or Passage 1? The answer is numbers. Freshly isolated cells are simply too few to treat a patient effectively. One or two rounds of expansion are necessary to get to a clinically useful dose.
This is where P2 and P3 come in. By the second or third passage, labs can typically produce enough cells to treat a patient without pushing the cells anywhere near the point where significant quality degradation has occurred. It is the point where quantity and quality are both acceptable at the same time.
P2 and P3 cells retain the genetic stability of early-stage cultures. They still have meaningful telomere length. Their surface markers are largely intact, meaning they are still expressing the proteins associated with therapeutic function. Their secretory profiles are still dominated by beneficial anti-inflammatory and tissue-supportive cytokines rather than the inflammatory mix that senescent cells produce. They maintain stronger differentiation potential, which matters particularly for stem cell applications where the goal involves guiding cells toward a specific tissue type.
For mesenchymal stem cells, which are among the most commonly used cells in regenerative medicine, the research literature consistently points to a performance peak in the P2 to P4 range, with a notable decline beyond that. The P2 and P3 window represents that peak for most donor sources and most clinical applications.
From a safety standpoint, early passage cells have had fewer opportunities to accumulate mutations. This is not a trivial concern. Regulatory agencies around the world pay close attention to passage number precisely because cells that have been divided many times carry a higher theoretical risk of having developed abnormal characteristics. Using P2 or P3 cells keeps the treatment well within the window where that risk is lowest.

The Problem with High-Passage Cells in Clinical Use
If the science so clearly favors early-passage cells, you might wonder why any clinic would use high-passage cells. The honest answer is that it often comes down to cost and logistics.
Maintaining a supply of low-passage cells requires either working with fresh tissue donations on a consistent basis or investing heavily in biobanking infrastructure that stores early-passage cells at scale. Both approaches are more expensive and operationally complex than simply continuing to expand an existing cell line through many passages to generate large quantities cheaply.
Highly passaged cells are also easier to stockpile for extended periods. A clinic that has banked P8 or P10 cells does not need to coordinate with tissue donors or maintain a rolling inventory of freshly expanded early-passage product. It can pull from a shelf and ship. That kind of operational convenience does not serve the patient, though. It serves the clinic’s logistics.
There is also a transparency problem. Unlike the concentration of a drug or the purity of a biological product, passage number is not something patients typically know to ask about. A clinic using P8 cells and a clinic using P2 cells can both describe their treatment as “mesenchymal stem cell therapy” without technically misrepresenting what they offer. The difference in the quality of what is being administered is enormous, but it is invisible to most patients.
High-passage cells also lose something that is hard to quantify but practically important: homing ability. When therapeutic cells are administered, they need to find their way to the areas of the body where they are needed. This is partly mediated by receptor proteins on the cell surface that help them respond to chemical signals from injured or inflamed tissue. High-passage cells often express lower levels of these homing receptors, meaning they are less able to migrate to where they could do the most good.

How Passage Affects Different Cell Types
The P2 to P3 principle holds broadly across cell types used in therapy, but it plays out differently depending on the specific cells involved.
For mesenchymal stem cells, which are derived from sources like bone marrow, umbilical cord tissue, or fat, the decline in key markers like CD73, CD90, and CD105 tends to become more pronounced after P5 or P6. Growth factor secretion, which is central to the tissue-supportive effects of MSC therapy, also drops significantly in later passages. The P2 to P3 window maintains high expression of these markers and strong secretory activity.
For immune cells like T cells and natural killer cells used in cancer-related applications, the concern takes a slightly different form. These cells can develop what researchers call an “exhaustion” phenotype with repeated expansion. Exhausted immune cells have reduced cytotoxic activity and are less capable of targeting diseased cells effectively. Early passage here means fewer cycles of artificial stimulation and expansion, which preserves the cells’ fighting capacity.
For tissue-specific cells like chondrocytes, which are used in cartilage repair applications, the risk of over-expansion is dedifferentiation. When chondrocytes are passaged too many times, they stop behaving like cartilage cells and start behaving more like generic connective tissue cells. They lose the collagen-producing and matrix-building properties that make them useful for joint repair. This transition can begin as early as P3 or P4 in some protocols, making early passage even more critical in this application.
The specifics vary by cell type, but the underlying principle is consistent: the more times cells have been copied, the further they are from their original identity and function.

Questions Every Patient Should Ask Before Treatment
Understanding passage number is only useful if it leads to action. If you are considering any kind of cell-based treatment, there are specific questions worth asking that can help you gauge whether a provider is working with high-quality product.
Start by asking directly: what passage number are the cells being used in my treatment? Any reputable clinic should be able to answer this. If they cannot, or if they seem unfamiliar with the question, that is a meaningful red flag.
Ask whether they can provide a Certificate of Analysis, or CoA, for the cells being used. A proper CoA includes information about passage number, viability, sterility testing, and often marker expression data. It is a standard quality control document in legitimate cell therapy, and its absence suggests inadequate quality oversight.
Ask how the clinic sources its cells and how recently they were expanded. Fresh expansion is preferable to cells that have been sitting in storage for an extended period, especially if passage number is already on the higher end.
Ask specifically how they define acceptable passage limits for clinical use, and what happens if a batch does not meet those criteria. A clinic that has clear internal standards and a process for rejecting out-of-spec batches is one that takes quality seriously. A clinic that cannot articulate those standards probably does not have them.
What Regulators and Research Institutions Say
Regulatory bodies including the FDA in the United States and the EMA in Europe have consistently recognized passage number as a relevant quality parameter in cell therapy manufacturing. Good Manufacturing Practice, or GMP, guidelines for cell therapy products include passage number as part of the identity and quality documentation that manufacturers are expected to track and report.
While there is no universal regulatory mandate that sets P2 or P3 as the hard upper limit for all cell types, the scientific consensus among researchers working in regenerative medicine is clear. Published studies on MSCs routinely use P2 to P4 cells for in vitro testing, specifically because cells at these passages are considered to reflect the properties that make the treatment relevant. Studies designed to characterize the therapeutic potential of MSCs generally do not use P8 or P10 cells, because the research community already understands that those cells do not represent the functional standard.
Major academic medical centers working on cell therapy research have established internal protocols that cap passage use at specific limits, often in the P3 to P5 range depending on cell type and application. These are not arbitrary choices. They reflect the accumulated weight of evidence showing that cell quality and potency decline with passage.
The Direction the Field Is Moving
The cell therapy industry has been developing tools and practices specifically to address the challenge of maintaining early-passage cell availability at the scale needed for clinical use.
Cryopreservation technology has improved significantly, making it possible to bank large quantities of P2 or P3 cells without significant damage to viability or function. This allows providers to freeze early-passage product, test it for quality, and release it for clinical use without needing to expand cells further. When done well, cryopreservation essentially allows a clinic to lock in the quality of early-passage cells and deploy them when needed.
Biobanking strategies are also advancing in ways that support early-passage use. Rather than expanding a single batch of cells through many passages over time, well-capitalized banks are working to maintain a rolling inventory of cells from multiple donors, keeping passage numbers low across their entire supply.
Gene expression profiling is beginning to be used as a quality control tool that goes beyond simple passage counting. Rather than relying solely on passage number as a proxy for quality, researchers can now run panels of gene expression markers to verify that cells are still displaying the functional identity associated with therapeutic potency. This kind of molecular quality control provides a more direct measure of whether cells are still in their optimal state, regardless of passage number.
Passage Number Is Not a Technicality
There is a tendency in medicine to treat quality control as a background concern, something labs and regulators worry about while patients and clinicians focus on outcomes. But the passage number is not a back-office detail. It is one of the core determinants of whether a cell therapy product has the biological capacity to do anything useful when administered.
The reason P2 and P3 cells are superior is not complicated. They are closer to the original tissue. They carry less accumulated damage. They express the proteins that make cell therapy work. They have the capacity to replicate, migrate, and communicate with surrounding tissue. They are not yet approaching the decline in function that higher-passage cells have already begun.
When you ask your provider about the passage number, you are not asking an overly technical question. You are asking about the quality of the product being put into your body. That is a completely reasonable thing to know, and any provider worth working with should welcome the question.
The best cell therapy programs in the world maintain strict passage controls not because they are required to on paper, but because the science makes it obvious. For patients, understanding why that matters puts you in a position to evaluate the clinics and treatments you are considering from an informed perspective rather than taking quality on faith.